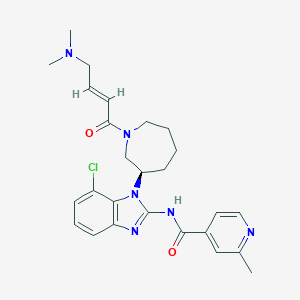
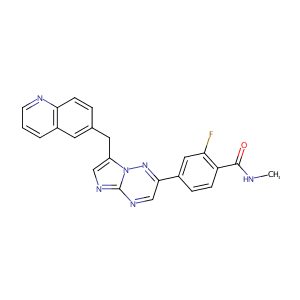
| 1 |
ClinicalTrials.gov (NCT02335944) Study of Safety and Efficacy of EGFR-TKI EGF816 in Combination With cMET Inhibitor INC280 in Adult Patients With EGFR Mutated Non Small Cell Lung Cancer.
|
| 2 |
Clinical pipeline report, company report or official report of the Pharmaceutical Research and Manufacturers of America (PhRMA)
|
| 3 |
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health Human Services. 2020
|
| 4 |
Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800031741)
|
| 5 |
Discovery of a mutant-selective covalent inhibitor of EGFR that overcomes T790M-mediated resistance in NSCLC. Cancer Discov. 2013 Dec;3(12):1404-15.
|
|
|
|
|
|
|