| 1 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
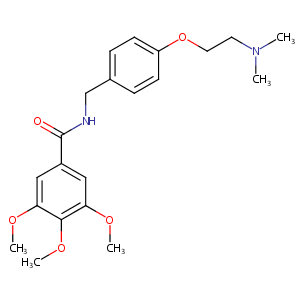
Trimethobenzamide FDA Label
|
| 3 |
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015
|
| 4 |
ClinicalTrials.gov (NCT02044861) Phase 1 Safety Study of ACT-PFK-158, 2HCl in Patients With Advanced Solid Malignancies. U.S. National Institutes of Health.
|
| 5 |
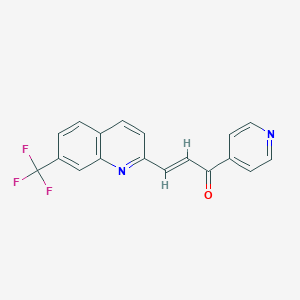
Discovery of a PFKFB3 inhibitor for phase I trial testing that synergizes with the B-Raf inhibitor vemurafenib. Cancer Metab. 2014; 2(Suppl 1): P14.
|
|
|
|
|
|
|