| 1 |
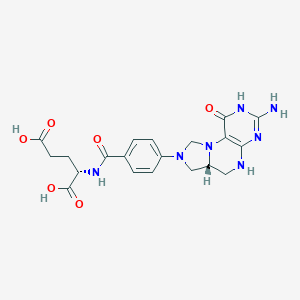
Loss of function mutations in VARS encoding cytoplasmic valyl-tRNA synthetase cause microcephaly, seizures, and progressive cerebral atrophy.Hum Genet. 2018 Apr;137(4):293-303. doi: 10.1007/s00439-018-1882-3. Epub 2018 Apr 24.
|
| 2 |
ClinicalTrials.gov (NCT03750786) A Study to Compare the Efficacy of Arfolitixorin Versus Leucovorin in Combination With 5 Fluorouracil, Oxaliplatin, and Bevacizumab in Patients With Advanced Colorectal Cancer (AGENT). U.S. National Institutes of Health.
|
| 3 |
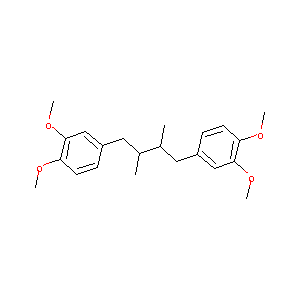
Terameprocol, a novel site-specific transcription inhibitor with anticancer activity. IDrugs. 2008 Mar;11(3):204-14.
|
|
|
|
|
|
|