Details of the Drug Combination
General Information of Drug Combination (ID: DCNMK6Y)
| Drug Combination Name |
Uridine triacetate Fludeoxyglucose F 18
|
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indication |
|
|||||||||||||||||
| Component Drugs | Uridine triacetate | Fludeoxyglucose F 18 | ||||||||||||||||
| Small molecular drug | Small molecular drug | |||||||||||||||||
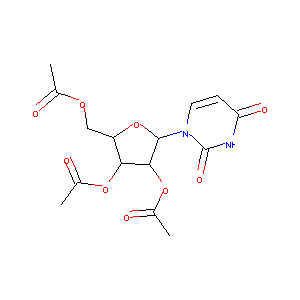
|
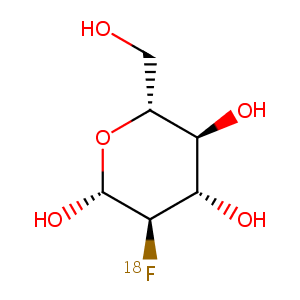
|
|||||||||||||||||
| 2D MOL | 2D MOL | |||||||||||||||||
| 3D MOL | 3D MOL | |||||||||||||||||
References
