Details of the Drug Combination
General Information of Drug Combination (ID: DCTMXIG)
| Drug Combination Name |
Terameprocol Crotamiton
|
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indication |
|
|||||||||||||||||
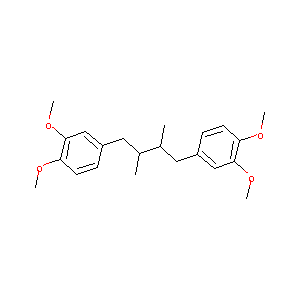
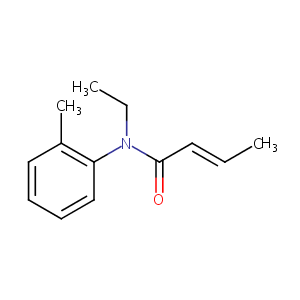
| Component Drugs | Terameprocol | Crotamiton | ||||||||||||||||
| Small molecular drug | Small molecular drug | |||||||||||||||||
|
|
|||||||||||||||||
| 2D MOL | 2D MOL | |||||||||||||||||
| 3D MOL | 3D MOL | |||||||||||||||||
| High-throughput Screening Result | Testing Cell Line: KBM-7 | |||||||||||||||||
| Zero Interaction Potency (ZIP) Score: 19.83 | ||||||||||||||||||
| Bliss Independence Score: 19.83 | ||||||||||||||||||
| Loewe Additivity Score: 24.65 | ||||||||||||||||||
| LHighest Single Agent (HSA) Score: 24.68 | ||||||||||||||||||
Molecular Interaction Atlas of This Drug Combination
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indication(s) of Terameprocol |
|
|||||||||||||||||||||||||
|
Terameprocol Interacts with 1 DTT Molecule(s)
|
||||||||||||||||||||||||||
| Indication(s) of Crotamiton |
|
|||||||||||||||||||||||||
References
