Details of the Drug
General Information of Drug (ID: DM1BYNF)
| Drug Name |
THRX-198321
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms | Beta 2 adrenoreceptor agonist/muscarinic receptor antagonist (asthma), Theravance | ||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Structure | 3D Structure is Not Available |
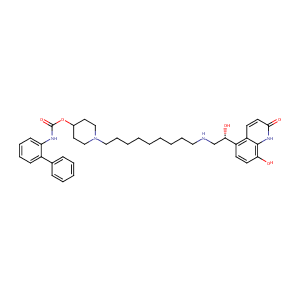 |
|||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| ICD Disease Classification | 12 Disease of the respiratory system | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Disease Class | ICD-11: CA23 Asthma | |||||||||||||||||||||||||||||
| The Studied Tissue | Lung tissue | |||||||||||||||||||||||||||||
| The Studied Disease | Chronic obstructive pulmonary disease [ICD-11:CA23] | |||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||
