Details of the Drug
General Information of Drug (ID: DM3LFDH)
| Drug Name |
AZD9977
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
AZD9977; AZD-9977; 1850385-64-6; BALCINRENONE; UNII-6C9UKZ0CYE; 6C9UKZ0CYE; Azd 9977; 2-[(3S)-7-Fluoro-4-(3-oxo-4H-1,4-benzoxazine-6-carbonyl)-2,3-dihydro-1,4-benzoxazin-3-yl]-N-methylacetamide; 2H-1,4-Benzoxazine-3-acetamide, 4-((3,4-dihydro-3-oxo-2H-1,4-benzoxazin-6-yl)carbonyl)-7-fluoro-3,4-dihydro-N-methyl-, (3S)-; 2-[(3~{S})-7-fluoranyl-4-[(3-oxidanylidene-4~{H}-1,4-benzoxazin-6-yl)carbonyl]-2,3-dihydro-1,4-benzoxazin-3-yl]-~{N}-methyl-ethanamide; 2-((3S)-7-FLUORO-4-((3-OXO-3,4-DIHYDRO-2H-1,4-BENZOXAZIN-6-YL)CARBONYL)-3,4-DIHYDRO-2H-1,4-BENZOXAZIN-3-YL)-N-METHYLACETAMIDE; 2-{(3S)-7-Fluoro-4-[(3-oxo-3,4-dihydro-2H-1,4-benzoxazin-6-yl)carbonyl]-3,4-dihydro-2H-1,4-benzoxazin-3-yl}-N-methylacetamide; BALCINRENONE [INN]; CHEMBL3916929; SCHEMBL17363859; GTPL11281; AZD 9977 [WHO-DD]; MBKYLPOPYYLTNW-ZDUSSCGKSA-N; BDBM238159; AKOS040759623; DB15418; US9394291, 4a; AC-36749; MS-26766; HY-120274; CS-0077469; F82308; (S)-1 [PMID: 30596500]; ECV
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecule
|
||||||||||||||||||||||
| Structure |
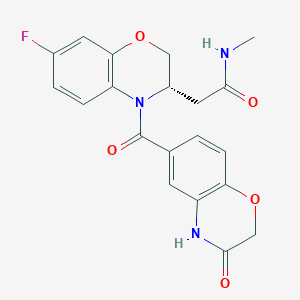 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Heart failure | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | BD10-BD13 | |||||||||||||||||||||||
|
||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||
References
