Details of the Drug
General Information of Drug (ID: DMA3DKO)
| Drug Name |
RIST4721
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
vimnerixin; AZD4721; RIST4721; AZD-4721; RIST-4721; X09ORN2BF0; 1418112-77-2; UNII-X09ORN2BF0; AZD-4721 [WHO-DD]; 1-Azetidinesulfonamide, N-(6-((1R,2S)-2,3-dihydroxy-1-methylpropoxy)-2-(((4-fluorophenyl)methyl)thio)-4-pyrimidinyl)-3-methyl-; vimnerixin [INN]; Azd 4721; CHEMBL5095217; SCHEMBL14579049; GTPL12100; AZD 4721 [WHO-DD]; AZD4721;RIST4721; AKOS040756752; compound 4 [WO2019055509A1]; MS-28748; HY-145640; CS-0377236; F83913; N-(6-{[(2R,3S)-3,4-dihydroxybutan-2-yl]oxy}-2-{[(4-fluorophenyl)methyl]sulfanyl}pyrimidin-4-yl)-3-methylazetidine-1-sulfonamide; N-[6-[(1R,2S)-2,3-Dihydroxy-1-methylpropoxy]-2-[[(4-fluorophenyl)methyl]thio]-4-pyrimidinyl]-3-methyl-1-azetidinesulfonamide; N-[6-[(2R,3S)-3,4-dihydroxybutan-2-yl]oxy-2-[(4-fluorophenyl)methylsulfanyl]pyrimidin-4-yl]-3-methylazetidine-1-sulfonamide
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| Drug Type |
Small molecule
|
||||||||||||||||||||||||||
| Structure |
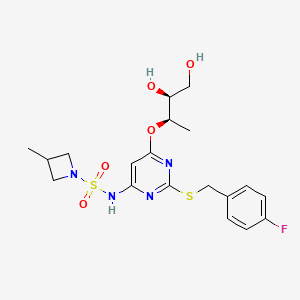 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Hidradenitis suppurativa | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | ED92.0 | |||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||
References
