Details of the Drug
General Information of Drug (ID: DMAD1S5)
| Drug Name |
UK-74505
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
rac-Modipafant; UK-74505; Modipafant racemate; 122956-68-7; UK 74505; 3-Pyridinecarboxylic acid, 4-(2-chlorophenyl)-1,4-dihydro-6-methyl-2-(4-(2-methyl-1H-imidazo(4,5-c)pyridin-1-yl)phenyl)-5-((2-pyridinylamino)carbonyl)-, ethyl ester; UK 80067; Modipafant (racemate); AC1L2W8B; C34H29ClN6O3; SCHEMBL6995473; AN-3660; LS-172547; KB-276046; FT-0672447; ethyl 4-(2-chlorophenyl)-6-methyl-2-[4-(2-methylimidazo[4,5-c]pyridin-1-yl)phenyl]-5-(2-pyridylcarbamoyl)-1,4-dihydropyridine-3-carboxylate; ethyl 4-(2-chlorophenyl)-6-methyl-2
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
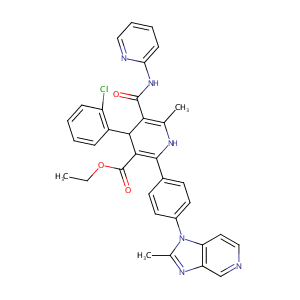 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 2 | Molecular Weight (mw) | 605.1 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 5.6 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 8 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 7 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Sepsis | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | 1G40-1G41 | |||||||||||||||||||||||
|
||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||
References
