Details of the Drug
General Information of Drug (ID: DMM4FJ3)
| Drug Name |
Finerenone
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
UNII-DE2O63YV8R; BAY 94-8862; 1050477-31-0; BAY94-8862; DE2O63YV8R; Finerenone [USAN:INN]; Finerenone (JAN/USAN/INN); SCHEMBL8157011; GTPL8678; DTXSID10146928; J3.584.878I; D10633; 1,6-Naphthyridine-3-carboxamide, 4-(4-cyano-2-methoxyphenyl)-5-ethoxy-1,4-dihydro-2,8-dimethyl-, (4S)-;1,6-Naphthyridine-3-carboxamide, 4-(4-cyano-2-methoxyphenyl)-5-ethoxy-1,4-dihydro-2,8-dimethyl-, (4S)-; 1,6-Naphthyridine-3-carboxamide, 4-(4-cyano-2-methoxyphenyl)-5-ethoxy-1,4-dihydro-2,8-dimethyl-, (4S)-; (4S)-4-(4-cyano-2-metho
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| Affected Organisms |
Humans
|
||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||
| Structure |
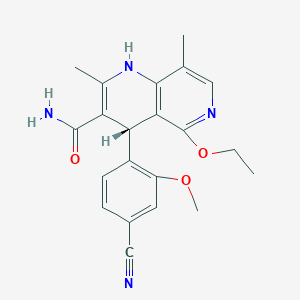 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 378.4 | |||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2.4 | ||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 5 | ||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | ||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Chronic kidney disease | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | GB61 | |||||||||||||||||||||||
|
||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||
References
