| Drug Name |
PT-302
|
| Synonyms |
149485-77-8; Thiourea, N-(2-(4-fluorophenyl)ethyl)-N'-2-thiazolyl-; Thiourea, N-[2-(4-fluorophenyl)ethyl]-N'-2-thiazolyl-; PETT Analog 4; AC1MHDMB; CHEMBL253996; BDBM1897; DTXSID00164292; 1-[2-(4-fluorophenyl)ethyl]-3-(1,3-thiazol-2-yl)thiourea; AKOS030030877; 1-(2-Thiazolyl)-3-[4-fluorophenethyl]thiourea; N-(2-(4-Fluorophenethyl))-N -(2-thiazolyl)thiourea; 1-[2-(4-fluorophenyl)ethyl]-3-thiazol-2-yl-thiourea
|
| Drug Type |
Small molecular drug
|
| Structure |
|
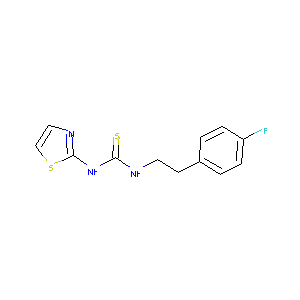 |
|
3D MOL
|
2D MOL
|
| #Ro5 Violations (Lipinski): 0 |
Molecular Weight (mw) |
281.4 |
|
| Logarithm of the Partition Coefficient (xlogp) |
2.9 |
| Rotatable Bond Count (rotbonds) |
4 |
| Hydrogen Bond Donor Count (hbonddonor) |
2 |
| Hydrogen Bond Acceptor Count (hbondacc) |
4 |
| Chemical Identifiers |
- Formula
- C12H12FN3S2
- IUPAC Name
1-[2-(4-fluorophenyl)ethyl]-3-(1,3-thiazol-2-yl)thiourea - Canonical SMILES
-
C1=CC(=CC=C1CCNC(=S)NC2=NC=CS2)F
- InChI
-
InChI=1S/C12H12FN3S2/c13-10-3-1-9(2-4-10)5-6-14-11(17)16-12-15-7-8-18-12/h1-4,7-8H,5-6H2,(H2,14,15,16,17)
- InChIKey
-
WGSDLRNUZBUXDV-UHFFFAOYSA-N
|
| Cross-matching ID |
- PubChem CID
- 3001164
- CAS Number
-
- TTD ID
- D03MLA
|
|
|
|
|
|
|
|

