Details of the Drug
General Information of Drug (ID: DMX8LY4)
| Drug Name |
NS-8
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
NS-8; 186033-14-7; 2-amino-3-cyano-4-methyl-5-(2-fluorophenyl)pyrrole; 2-amino-5-(2-fluorophenyl)-4-methyl-1H-pyrrole-3-carbonitrile; KVDWOQGDMAEJLM-UHFFFAOYSA-N; CHEMBL42661; SCHEMBL4624842; ZINC21886; MolPort-005-943-258; AKOS027201108; EN300-254366; 2-amino-3-cyano-4-methyl-5-(2fluorophenyl)pyrrole; Z2306534448
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
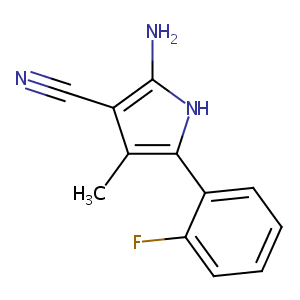 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 215.23 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2.8 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 1 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
References
