Details of the Drug
General Information of Drug (ID: DMYCV7U)
| Drug Name |
N,O6-Disulfo-Glucosamine
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
N,O6-Disulfo-Glucosamine; 2-deoxy-6-O-sulfo-2-(sulfoamino)-alpha-D-glucopyranose; SGN; AC1L9G8A; SCHEMBL1009127; DB03959; 6-O-sulfo-N-sulfo-alpha-d-glucosamine; J3.578.180C; 6-O-Sulfo-2-(sulfoamino)-2-deoxy-alpha-D-glucopyranose; [(2S,3R,4R,5S,6R)-2,4,5-trihydroxy-6-(sulfooxymethyl)oxan-3-yl]sulfamic acid; WURCS=2.0/1,1,0/[a2122h-1a_1-5_2*NSO/3=O/3=O_6*OSO/3=O/3=O]/1/
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
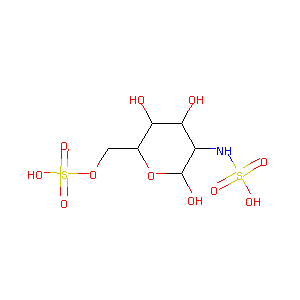 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 3 | Molecular Weight (mw) | 339.3 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | -4.2 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 5 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 6 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 12 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Discovery agent | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | N.A. | |||||||||||||||||||||||
|
||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||
