Details of the Drug
General Information of Drug (ID: DMZRXTL)
| Drug Name |
E7820
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
E7820; 289483-69-8; E-7820; E 7820; UNII-TVH5K7949N; TVH5K7949N; 3-cyano-N-(3-cyano-4-methyl-1H-indol-7-yl)benzenesulfonamide; N-(3-cyano-4-methyl-1H-indol-7-yl)-3-cyanobenzene-sulfonamide; NSC-719239; N-(3-cyano-4-methyl-1H-indol-7-yl)-3-cyanobenzenesulfonamide; Benzenesulfonamide, 3-cyano-N-(3-cyano-4-methyl-1H-indol-7-yl)-; LWGUASZLXHYWIV-UHFFFAOYSA-N; AC1L52N5; ZINC8704; SCHEMBL1581157; DTXSID20183142; BCP25835; EX-A1818; 3-Cyano-N-(3-cyano-4-methyl-1H-indole-7-yl)benzensulfonamide; NSC719239; AKOS032944953; CS-6075
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||
| Structure |
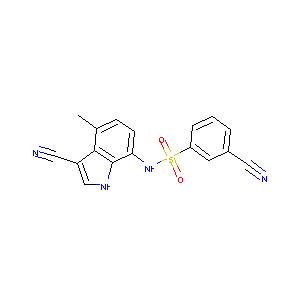 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 336.4 | |||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2.4 | ||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 3 | ||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | ||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
References
