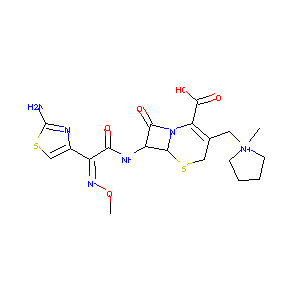
| Synonyms |
Axepim; CFPM; Cefepima; Cefepimum; Cepimax; Cepimex; Maxcef; Maxipime; Cefepima [Spanish]; Cefepimum [Latin]; BMY 28142; Axepim (TN); BMY-28142; Cefepime [USAN:INN]; Cepimax (TN); Cepimex (TN); Maxcef (TN); Maxipime (TN); Cefepime (USAN/INN); (6R,7R)-7-[[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-methoxyiminoacetyl]amino]-3-[(1-methylpyrrolidin-1-ium-1-yl)methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate; (6R,7R)-7-{[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-(methoxyimino)acetyl]amino}-3-[(1-methylpyrrolidinium-1-yl)methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate; 1-(((6R,7R)-7-(2-(2-Amino-4-thiazolyl)glyoxylamido)-2-carboxy-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-en-3-yl)methyl)-1-methylpyrrolidinium hydroxide, inner salt, 7(sup 2)-(Z)-(O-methyloxime); 7beta-[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-(methoxyimino)acetamido]-3-[(1-methylpyrrolidinium-1-yl)methyl]-3,4-didehydrocepham-4-carboxylate
|