Details of the Drug
General Information of Drug (ID: DMWJE9D)
| Drug Name |
Ambroxol
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Ambroxol hydrochloride; 23828-92-4; Ambroxol HCl; Mucosolvan; Mucoangin; Ambroxolhydrochloride; cis-4-((2-Amino-3,5-dibromobenzyl)amino)cyclohexanol hydrochloride; UNII-CC995ZMV90; Ambroxol hydrochloride; trans-4-((2-Amino-3,5-dibromobenzyl)amino)cyclohexanol hydrochloride; Ambroxol Hydrochloride [JAN]; CC995ZMV90; 15942-05-9; Ambroxol hydrochloride (JAN); 2-Amino-3,5-dibromo-N-[trans-4-hydroxycyclohexyl]benzylamine; 4-((2-amino-3,5-dibromobenzyl)amino)cyclohexan-1-ol hydrochloride
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||
| Structure |
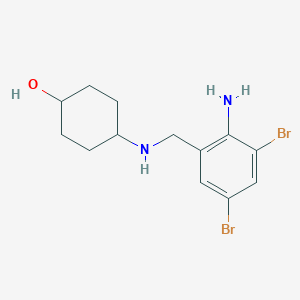 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 378.1 | |||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2.6 | ||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 3 | ||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | ||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 3 | ||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug-Metabolizing Enzyme (DME) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
References
