| 1 |
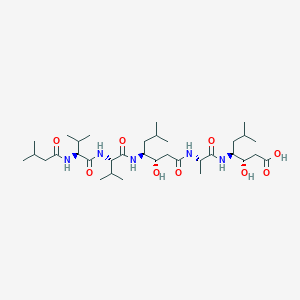
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800004280)
|
| 3 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 953).
|
| 4 |
Novel molecular targets for antimalarial drug development. Chem Biol Drug Des. 2008 Apr;71(4):287-97.
|
| 5 |
Novel molecular targets for antimalarial chemotherapy. Int J Antimicrob Agents. 2007 Jul;30(1):4-10.
|
| 6 |
Evidence of a lysosomal pathway for apoptosis induced by the synthetic retinoid CD437 in human leukemia HL-60 cells. Cell Death Differ. 2001 May;8(5):477-85. doi: 10.1038/sj.cdd.4400843.
|
| 7 |
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services.
|
| 8 |
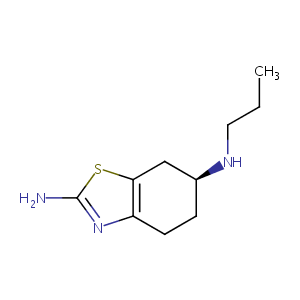
Uptake of pramipexole by human organic cation transporters. Mol Pharm. 2010 Aug 2;7(4):1342-7.
|
| 9 |
OCT1 polymorphism is associated with response and survival time in anti-Parkinsonian drug users. Neurogenetics. 2011 Feb;12(1):79-82.
|
|
|
|
|
|
|