| 1 |
ClinicalTrials.gov (NCT02401880) Effects of Linagliptin in Addition to Empagliflozin on Islet Cell Physiology
|
| 2 |
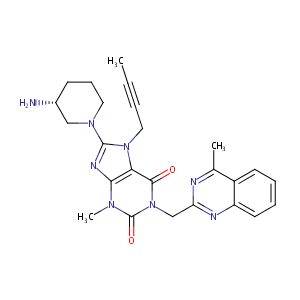
Linagliptin FDA Label
|
| 3 |
FDA Approved Drug Products from FDA Official Website. 2019. Application Number: (NDA) 208026
|
| 4 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6318).
|
| 5 |
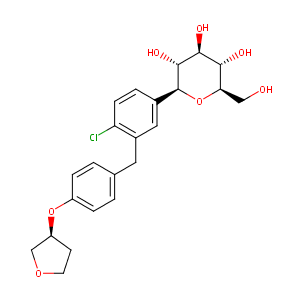
ClinicalTrials.gov (NCT04371978) Efficacy and Safety of Dipeptidyl Peptidase-4 Inhibitors in Diabetic Patients With Established COVID-19. U.S. National Institutes of Health.
|
| 6 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 4754).
|
| 7 |
Boehringer Ingelheim. Product Development Pipeline. June 2 2009.
|
| 8 |
DPP4 inhibition: preventing SARS-CoV-2 infection and/or progression of COVID-19 Diabetes Metab Res Rev. 2020 Apr 26.
|
| 9 |
Tarascon Pocket Pharmacopoeia 2018 Classic Shirt-Pocket Edition.
|
| 10 |
2014 FDA drug approvals. Nat Rev Drug Discov. 2015 Feb;14(2):77-81.
|
| 11 |
KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 2017 Jan 4;45(D1):D353-D361. (dg:DG01665)
|
| 12 |
KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 2017 Jan 4;45(D1):D353-D361. (dg:DG01913)
|
| 13 |
Empagliflozin (Jardiance): a novel SGLT2 inhibitor for the treatment of type-2 diabetes. P T. 2015 Jun;40(6):364-8.
|
| 14 |
ClinicalTrials.gov (NCT01734785) Safety and Efficacy of the Combination of Empagliflozin and Linagliptin Compared to Linagliptin Alone Over 24 Weeks in Patients With Type 2 Diabetes. U.S. National Institutes of Health.
|
|
|
|
|
|
|