| 1 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015
|
| 3 |
FDA Approved Drug Products from FDA Official Website. 2009. Application Number: (NDA) 018749.
|
| 4 |
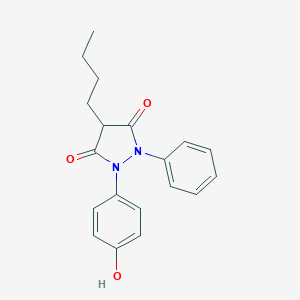
Effect of albumin conformation on binding of phenylbutazone and oxyphenbutazone to human serum albumin. J Pharm Sci. 1982 Feb;71(2):241-4.
|
| 5 |
A Gene Expression Biomarker Predicts Heat Shock Factor 1 Activation in a Gene Expression Compendium. Chem Res Toxicol. 2021 Jul 19;34(7):1721-1737. doi: 10.1021/acs.chemrestox.0c00510. Epub 2021 Jun 25.
|
| 6 |
Enzymatic, immunological and phylogenetic characterization of Brucella suis urease. BMC Microbiol. 2008 Jul 19;8:121.
|
| 7 |
New bispyridinium oximes: in vitro and in vivo evaluation of their biological efficiency in soman and tabun poisoning. Chem Biol Interact. 2008 Sep 25;175(1-3):413-6.
|
|
|
|
|
|
|