Details of the Drug Combinations
General Information of This Drug (ID: DMQ9MVG)
| Drug Name | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
AVAGARD; Chlorhexidin; Chlorhexidinum; Cloresidina; Clorhexidina; Fimeil; Hexadol; Hibiclens; Hibispray; Hibistat;Nolvasan; Novalsan; Peridex; Periogard; Rotersept; Savloclens; Soretol; Sterido; Sterilon; Superspray; Tubulicid; Chlorhexidin [Czech]; Chlorhexidine Base; Chlorhexidine gluconate; Cloresidina [DCIT]; Decanoylacetaldehyde Sodium Sulfide; Dentisept [veterinary]; Savlon babycare; Sebidin A; Sodium Houttuyfonamide; Avagard (TN); BIOPATCH (TN); ChloraPrep (TN); Chlorhexamed (TN); Chlorhexidine (INN);Chlorhexidine [INN:BAN]; Chlorhexidinum [INN-Latin]; Chlorohex (TN); Clohex (TN); Clorexil (TN); Clorhexidina [INN-Spanish]; Corsodyl (TN); Dentisept [veterinary] (TN); Dexidin (TN); Exidine (TN); Hibiclens (TN); Hibiscrub (TN); MK-412A; Merfen-incolore; Oro-Clense; Oronine (TN); Perichlor (TN); Peridex (TN); Peridont (TN); Periochip (TN); Periogard Oral Rinse (TN); Perioxidina (TN); Savacol (TN); Suthol (TN); Dejavu-MW (TN); Merfen-incolore (TN); Lisium (*Dihydrochloride*); Nolvasan (*Diacetate*); SOLU-I.V (TN); N,N'-Bis(4-chlorophenyl)-3,12-diimino-2,4,11,13-tetraazatetradecanediimidamide; N,N''''-hexane-1,6-diylbis[N'-(4-chlorophenyl)(imidodicarbonimidic diamide)]; N,N'-Bis(4-chlorophenyl)-3,12-diimino-2,4,11,13-tetraazatetradeca-nediimidamide; N',N'''''-hexane-1,6-diylbis[N-(4-chlorophenyl)(imidodicarbonimidic diamide)]; 1,1 inverted exclamation marka-Hexamethylenebis[5-(4-chlorophenyl)biguanide]; 1,1'-Hexamethylene bis(5-(p-chlorophenyl)biguanide); 1,1'-Hexamethylenebis(5-(p-chlorophenyl)biguanide); 1,1'-Hexamethylenebis[5-(4-chlorophenyl)biguanide; 1,6-Bis(5-(p-chlorophenyl)biguandino)hexane; 1,6-Bis(p-chlorophenyldiguanido)hexane; 1,6-Di(4'-chlorophenyldiguanido)hexane; 1,6-Di(N-p-chlorophenyldiguanido)hexane
|
||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||
| Therapeutic Class |
Antibiotics
|
||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||
| Structure |
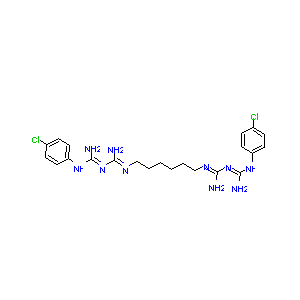 |
||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||
List of Combinatorial Drugs (CBD) Containing This Drug
|
5 Clinical Trial Drug Combination(s) Consisting of This drug
|
|||||||||||||||||||||||||||||||||||||||||||||
References
