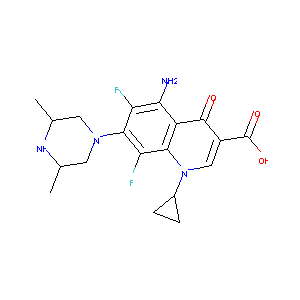
| Synonyms |
Esparfloxacino; SPFX; Spara; Sparfloxacine; Sparfloxacinum; Zagam; AT 4140; CP 103826; PD 131501; PD131501; AT-4140; CP-103826; DRG-0143; Esparfloxacino [INN-Spanish]; Liposome-encapsulated sparfloxacin; PD 1315-1; PD-131501; RP-64206; Respipac (TN); Sparfloxacin & RU 40555; Sparfloxacine [INN-French]; Sparfloxacinum [INN-Latin]; Zagam (TN); Sparfloxacin, cis-isomer; Sparfloxacin (JAN/USAN/INN); Sparfloxacin [USAN:BAN:INN:JAN]; Cis-5-Amino-1-cyclopropyl-7-(3,5-dimethyl-1-piperazinyl)-6,8-difluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic acid; (cis)-5-amino-1-cyclopropyl-7-(3,5-dimethyl-1-piperazinyl)-6,8-difluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic acid; 5-Amino-1-cyclohexyl-7-(cis-3,5-dimethylpiperazino)-6,8-difluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic acid; 5-Amino-1-cyclopropyl-7-(cis-3,5-dimethyl)-6,8-difluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic acid & RU 40555; 5-Amino-1-cyclopropyl-7-(cis-3,5-dimethyl-1-piperazinyl)-6,8-difluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic acid; 5-amino-1-cyclopropyl-7-[(3R,5S)-3,5-dimethylpiperazin-1-yl]-6,8-difluoro-4-oxo-1,4-dihydroquinoline-3-carboxylic acid; 5-amino-1-cyclopropyl-7-[(3R,5S)-3,5-dimethylpiperazin-1-yl]-6,8-difluoro-4-oxoquinoline-3-carboxylic acid
|