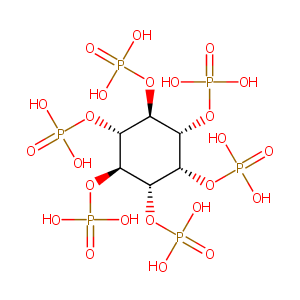
| Synonyms |
Phytic acid; 83-86-3; Phytate; Fytic acid; Inositol hexaphosphate; Alkalovert; myo-Inositol hexaphosphate; myo-inositol hexakisphosphate; Alkovert; Phytine; Acidum fyticum; Acide fytique; Acido fitico; myo-Inositol, hexakis(dihydrogen phosphate); myo-Inosistol hexakisphosphate; inositol hexakisphosphate; Inositol 1,2,3,4,5,6-hexakisphosphate; CCRIS 4513; UNII-7IGF0S7R8I; Saure des phytins [German]; Saeure des phytins; Acidum fyticum [INN-Latin]; Acido fitico [INN-Spanish]; Acide fytique [INN-French]; Inosithexaphosphorsaure; Phytate; SNF-03; SNF-04; SNF-471; SNF-472; SNF-571; SNF-671; Calcium metabolism modulator (transdermal patch formulation, cardiovascular calcifications), Sanifit Laboratoris SL; InsP6 (transdermal patch formulation, cardiovascular calcifications), Sanifit Laboratoris SL; Phytate (intravenous formulation, cardiovascular calcifications), Sanifit Laboratoris SL; Phytate (tablet formulation, osteoporosis), Sanifit Laboratoris SL; Phytate (transdermal patch formulation, cardiovascular calcifications), Sanifit Laboratoris SL; Calcium metabolism modulator (subcutaneous/injectable formulation, cardiovascular calcifications), Sanifit Laboratoris SL; Myo-inositol hexakisphosphate (transdermal patchformulation, cardiovascular calcifications), Sanifit Laboratoris SL; Myo-inositol hexaphosphate (intravenous formulation, cardiovascular calcifications), Sanifit Laboratoris SL; Myo-inositol hexaphosphate (tablet formulation, osteoporosis), Sanifit Laboratoris SL; Myo-inositol hexaphosphate (transdermal patch formulation, cardiovascular calcifications), Sanifit Laboratoris SL; Phytate (transdermal/patch formulation, renal calcium lithiasis), Sanifit Laboratoris SL; SNF-471 (transdermal patch, cardiovascular calcification), Sanifit; SNF-571 (transdermal patch, renal calcium lithiasis), Sanifit; InsP6, (transdermal/patch formulation, renal calcium lithiasis), Sanifit Laboratoris SL; Myo-inositol hexaphosphate (transdermal/patch formulation, renal calcium lithiasis), Sanfit Laboratoris SL; RP-3000
|