Details of the Drug
General Information of Drug (ID: DMDCFXJ)
| Drug Name |
Vaborbactam
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
RPX-7009; 1360457-46-0; RPX7009; UNII-1C75676F8V; 2-((3R,6S)-2-hydroxy-3-(2-(thiophen-2-yl)acetamido)-1,2-oxaborinan-6-yl)acetic acid; CHEMBL3317857; 1C75676F8V; 2-[(3R,6S)-2-hydroxy-3-[(2-thiophen-2-ylacetyl)amino]oxaborinan-6-yl]acetic acid; Vaborbactam [INN]; RPX 7009; Vaborbactam. RPX7009; Vaborbactam (USAN/INN); Vaborbactam [USAN:INN]; SCHEMBL620289; GTPL10871; Vabomere (vaborbactam + meropenem); EX-A2589; BDBM50089084; MFCD28502176; AKOS032961376; CS-6445; DB12107; SB17184; 1,2-Oxaborinane-6-acetic acid, 2-hydroxy-3-((2-(2-thienyl)acetyl)amino)-, (3R,6S)-; compound 9f [PMID: 25782055]; NCGC00510003-01; HY-19930; 2-hydroxy-3-((2-(2-thienyl)acetyl)amino)-; D10998; Q27252228; {(3R,6S)-2-hydroxy-3-[(thiophen-2-ylacetyl)amino]-1,2-oxaborinan-6-yl}acetic acid; 2-((3R,6S)-2-hydroxy-3-(2-(thiophen-2-yl)acetamido)-1,2-oxaborinan-6-yl)acetic acid; 2-[(3R,6S)-2-hydroxy-3-[(2-thiophen-2-ylacetyl)amino]oxaborinan-6-yl]acetic acid; 1,2-Oxaborinane-6-acetic acid
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Affected Organisms |
Escherichia coliKlebsiella pneumoniaeEnterobacter cloacae
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
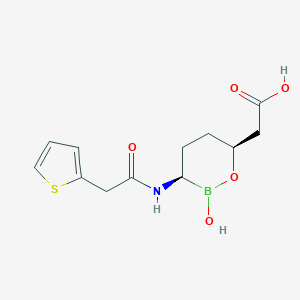 |
||||||||||||||||||||||
| 3D MOL is unavailable | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 |
Molecular Weight | 297.14 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient | Not Available | ||||||||||||||||||||||
| Rotatable Bond Count | 5 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count | 3 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count | 6 | ||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
References
