Details of the Drug
General Information of Drug (ID: DMG9HIA)
| Drug Name |
Urofollitropin
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Bravelle; Fertinex; Metrodin; Urofollitrophin; Fertinorm HP; Bravelle (TN); Fertinex (TN); Follistim (TN); Gonal-F(TN); (4-Threonine)oxytocin; 1,2-Dithia-5,8,11,14,17-penaazacycloeicosane, cyclic peptide deriv.; 4-(L-Threonine)oxytocin
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Therapeutic Class |
Fertility Agents
|
||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Sequence |
>Alpha chain
APDVQDCPECTLQENPFFSQPGAPILQCMGCCFSRAYPTPLRSKKTMLVQKNVTSESTCC VAKSYNRVTVMGGFKVENHTACHCSTCYYHKS >Beta chain NSCELTNITIAIEKEECRFCISINTTWCAGYCYTRDLVYKDPARPKIQKTCTFKELVYET VRVPGCAHHADSLYTYPVATQCHCGKCDSDSTDCTVRGLGPSYCSFGEMKE |
||||||||||||||||||||||
| Structure |
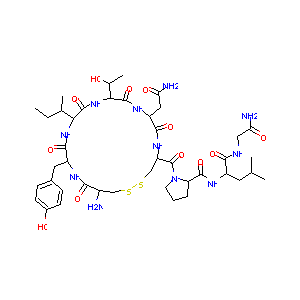 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 4 | Molecular Weight (mw) | 980.2 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | -1.5 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 15 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 12 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 15 | ||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Female infertility | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | GA31.Z | |||||||||||||||||||||||
|
||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | Natural products as sources of new drugs over the last 25 years. J Nat Prod. 2007 Mar;70(3):461-77. | ||||
|---|---|---|---|---|---|
| 2 | Trend Analysis of a Database of Intravenous Pharmacokinetic Parameters in Humans for 1352 Drug Compounds | ||||
| 3 | Follicle-stimulating hormone in clinical practice: an update. Treat Endocrinol. 2004;3(3):161-71. | ||||
