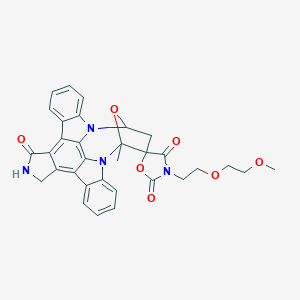
| Chemical Identifiers |
- Formula
- C32H28N4O7
- IUPAC Name
3-[2-(2-methoxyethoxy)ethyl]-15'-methylspiro[1,3-oxazolidine-5,16'-28-oxa-4,14,19-triazaoctacyclo[12.11.2.115,18.02,6.07,27.08,13.019,26.020,25]octacosa-1,6,8,10,12,20,22,24,26-nonaene]-2,3',4-trione - Canonical SMILES
-
CC12C3(CC(O1)N4C5=CC=CC=C5C6=C7C(=C8C9=CC=CC=C9N2C8=C64)CNC7=O)C(=O)N(C(=O)O3)CCOCCOC
- InChI
-
InChI=1S/C32H28N4O7/c1-31-32(29(38)34(30(39)43-32)11-12-41-14-13-40-2)15-22(42-31)35-20-9-5-3-7-17(20)24-25-19(16-33-28(25)37)23-18-8-4-6-10-21(18)36(31)27(23)26(24)35/h3-10,22H,11-16H2,1-2H3,(H,33,37)
- InChIKey
-
MTDVEOMKVDOULD-UHFFFAOYSA-N
|