Details of the Drug
General Information of Drug (ID: DMQZ5HF)
| Drug Name |
KNI-272
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
NSC651714; NSC-651714; AC1Q5L2H; (4r)-n-tert-butyl-3-[2-hydroxy-3-({n-[(isoquinolin-5-yloxy)acetyl]-s-methyl-l-cysteinyl}amino)-4-phenylbutanoyl]-1,3-thiazolidine-4-carboxamide; AC1L88B1; KNI 272; 3-(3,4-Dimethoxyphenyl)propionamide; NCI60_018063; 4-Thiazolidinecarboxamide,1-dimethylethyl)-3- [2-hydroxy-3-[[2-[[(5-isoquinolinyloxy)acetyl]amino]-3- (methylthio)-1-oxopropyl]amino]-1-oxo-4-phenylbutyl]-,[4R-[3[2S*,3S*(R*)],4R*]]
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
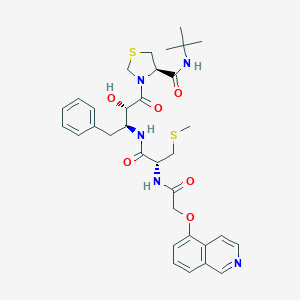 |
||||||||||||||||||||||
| 3D MOL is unavailable | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 2 | Molecular Weight (mw) | 667.8 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 3.2 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 14 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 4 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 9 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug-Metabolizing Enzyme (DME) |
|
||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
References
