Details of the Drug
General Information of Drug (ID: DMUW6QX)
| Drug Name |
E-6123
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
E 6123; 131614-02-3; E-6123; BRN 4280434; E- 6123; 4H-Pyrido(4',3':4,5)thieno(3,2-f)(1,2,4)triazolol(4,3-a)(1,4)diazepine, 6-(2-chlorophenyl)-9-(cyclopropylcarbonyl)-7,8,9,10-tetrahydro-1,4-dimethyl-, (S)-; 4H-Pyrido(4',3':4,5)thieno(3,2-f)(1,2,4)triazolo(4,3-a)(1,4)diazepine, 7,8,9,10-tetrahydro-6-(2-chlorophenyl)-9-(cyclopropylcarbonyl)-1,4-dimethyl-, (S)-; AC1MHYY4; CHEMBL114014; DTXSID20157146; API0009208; HY-10164; LS-134356
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
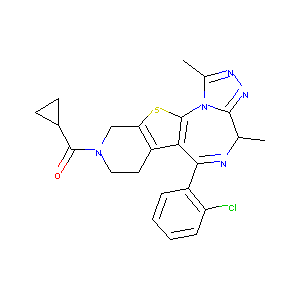 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 452 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 3.5 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 2 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 0 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Asthma | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | CA23 | |||||||||||||||||||||||
|
||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||
References
