Details of the Drug
General Information of Drug (ID: DMXH2R3)
| Drug Name |
CGP 77675
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
234772-64-6; CGP-77675; UNII-EQH27E0WRV; 1-(4-(4-amino-5-(3-methoxyphenyl)-7H-pyrrolo[2,3-d]pyrimidin-7-yl)phenethyl)piperidin-4-ol; CGP77675; EQH27E0WRV; CHEMBL475584; 1-[2-[4-[4-amino-5-(3-methoxyphenyl)pyrrolo[2,3-d]pyrimidin-7-yl]phenyl]ethyl]piperidin-4-ol; 4-Piperidinol, 1-(2-(4-(4-amino-5-(3-methoxyphenyl)-7H-pyrrolo(2,3-d)pyrimidin-7-yl)phenyl)ethyl)-; 4-piperidinol, 1-[2-[4-[4-amino-5-(3-methoxyphenyl)-7h-pyrrolo[2,3-d]pyrimidin-7-yl]phenyl]ethyl]-; AC1NSKIU; SCHEMBL6250604; CTK8E3165; DTXSID20178033
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
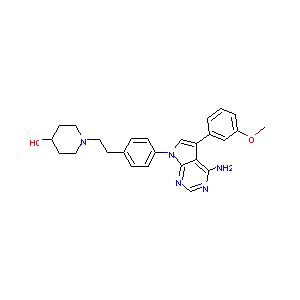 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 443.5 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 3.7 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 6 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 6 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Discovery agent | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | N.A. | |||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||
References
