| 1 |
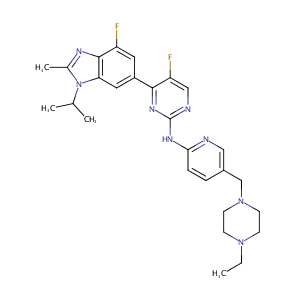
ClinicalTrials.gov (NCT02057133) A Study of LY2835219 (Abemaciclib) in Combination With Therapies for Breast Cancer That Has Spread
|
| 2 |
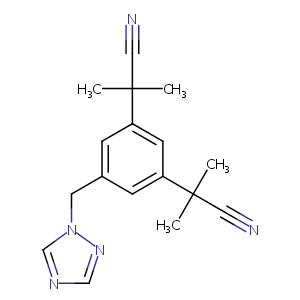
2017 FDA drug approvals.Nat Rev Drug Discov. 2018 Feb;17(2):81-85.
|
| 3 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7382).
|
| 4 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 5137).
|
| 5 |
Anastrozole FDA Label
|
| 6 |
Biological specificity of CDK4/6 inhibitors: dose response relationship, in vivo signaling, and composite response signature. Oncotarget. 2017 Jul 4;8(27):43678-43691. doi: 10.18632/oncotarget.18435.
|
| 7 |
Interpreting expression profiles of cancers by genome-wide survey of breadth of expression in normal tissues. Genomics 2005 Aug;86(2):127-41.
|
| 8 |
Abemaciclib Inhibits Renal Tubular Secretion Without Changing Glomerular Filtration Rate. Clin Pharmacol Ther. 2019 May;105(5):1187-1195.
|
| 9 |
LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]-Abemaciclib.
|
| 10 |
CDK4/6 Inhibition Controls Proliferation of Bladder Cancer and Transcription of RB1. J Urol. 2016 Mar;195(3):771-9. doi: 10.1016/j.juro.2015.08.082. Epub 2015 Aug 28.
|
| 11 |
Effect of abemaciclib (LY2835219) on enhancement of chemotherapeutic agents in ABCB1 and ABCG2 overexpressing cells in vitro and in vivo. Biochem Pharmacol. 2017 Jan 15;124:29-42. doi: 10.1016/j.bcp.2016.10.015. Epub 2016 Nov 2.
|
| 12 |
In vitro therapeutic effects of abemaciclib on triple-negative breast cancer cells. J Biochem Mol Toxicol. 2021 Sep;35(9):e22858. doi: 10.1002/jbt.22858. Epub 2021 Jul 26.
|
| 13 |
Effective aromatase inhibition by anastrozole in a patient with gonadotropin-independent precocious puberty in McCune-Albright syndrome. J Pediatr Endocrinol Metab. 2002;15 Suppl 3:945-8.
|
| 14 |
Anastrozole Aromatase Inhibitor Plasma Drug Concentration Genome-Wide Association Study: Functional Epistatic Interaction Between SLC38A7 and ALPPL2. Clin Pharmacol Ther. 2019 Jan 16.
|
| 15 |
In vitro and in vivo oxidative metabolism and glucuronidation of anastrozole. Br J Clin Pharmacol. 2010 Dec;70(6):854-69.
|
| 16 |
ADReCS-Target: target profiles for aiding drug safety research and application. Nucleic Acids Res. 2018 Jan 4;46(D1):D911-D917. doi: 10.1093/nar/gkx899.
|
| 17 |
Aromatase inhibitors: cellular and molecular effects. J Steroid Biochem Mol Biol. 2005 May;95(1-5):83-9. doi: 10.1016/j.jsbmb.2005.04.010.
|
| 18 |
Aromatase inhibition: translation into a successful therapeutic approach. Clin Cancer Res. 2005 Apr 15;11(8):2809-21. doi: 10.1158/1078-0432.CCR-04-2187.
|
| 19 |
Assessment of Five Pesticides as Endocrine-Disrupting Chemicals: Effects on Estrogen Receptors and Aromatase. Int J Environ Res Public Health. 2022 Feb 10;19(4):1959. doi: 10.3390/ijerph19041959.
|
| 20 |
Inhibition of estrogen receptor reduces connexin 43 expression in breast cancers. Toxicol Appl Pharmacol. 2018 Jan 1;338:182-190. doi: 10.1016/j.taap.2017.11.020. Epub 2017 Nov 24.
|
| 21 |
Biomarker changes during neoadjuvant anastrozole, tamoxifen, or the combination: influence of hormonal status and HER-2 in breast cancer--a study from the IMPACT trialists. J Clin Oncol. 2005 Apr 10;23(11):2477-92. doi: 10.1200/JCO.2005.07.559. Epub 2005 Mar 14.
|
| 22 |
ClinicalTrials.gov (NCT02441946) A Neoadjuvant Study of Abemaciclib (LY2835219) in Postmenopausal Women With Hormone Receptor Positive, HER2 Negative Breast Cancer
|
|
|
|
|
|
|