| 1 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6798).
|
| 3 |
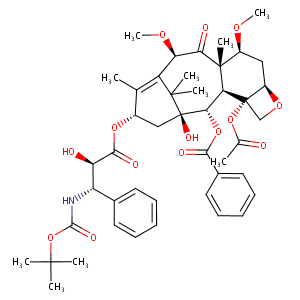
Cabazitaxel FDA Label
|
| 4 |
Lenalidomide FDA Label
|
| 5 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7331).
|
| 6 |
Low-dose Lenalidomide for Non-severe COVID-19 Treatment Trial (GETAFE)
|
| 7 |
Mullard A: 2010 FDA drug approvals. Nat Rev Drug Discov. 2011 Feb;10(2):82-5.
|
| 8 |
DAILYMED.nlm.nih.gov: JEVTANA- cabazitaxel kit.
|
| 9 |
The triphenyltin carboxylate derivative triphenylstannyl 2-(benzylcarbamoyl)benzoate impedes prostate cancer progression via modulation of Akt/FOXO3a signaling. Toxicol Appl Pharmacol. 2020 Aug 15;401:115091. doi: 10.1016/j.taap.2020.115091. Epub 2020 Jun 7.
|
| 10 |
Thalidomide and thalidomide analogues for maintenance of remission in Crohn's disease. Cochrane Database Syst Rev. 2009 Apr 15;(2):CD007351.
|
| 11 |
DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res. 2018 Jan 4;46(D1):D1074-D1082. (ID: DB00480)
|
| 12 |
Thalidomide and Its Analogs Differentially Target Fibroblast Growth Factor Receptors: Thalidomide Suppresses FGFR Gene Expression while Pomalidomide Dampens FGFR2 Activity. Chem Res Toxicol. 2019 Apr 15;32(4):589-602. doi: 10.1021/acs.chemrestox.8b00286. Epub 2019 Mar 15.
|
| 13 |
Circulating endothelial progenitor cells in multiple myeloma: implications and significance. Blood. 2005 Apr 15;105(8):3286-94. doi: 10.1182/blood-2004-06-2101. Epub 2004 Dec 23.
|
| 14 |
Structure of the human Cereblon-DDB1-lenalidomide complex reveals basis for responsiveness to thalidomide analogs. Nat Struct Mol Biol. 2014 Sep;21(9):803-9. doi: 10.1038/nsmb.2874. Epub 2014 Aug 10.
|
| 15 |
Thalidomide promotes degradation of SALL4, a transcription factor implicated in Duane Radial Ray syndrome. Elife. 2018 Aug 1;7:e38430. doi: 10.7554/eLife.38430.
|
| 16 |
A Dual Color Immunohistochemistry Assay for Measurement of Cereblon in Multiple Myeloma Patient Samples. Appl Immunohistochem Mol Morphol. 2016 Nov/Dec;24(10):695-702. doi: 10.1097/PAI.0000000000000246.
|
| 17 |
Biologically active neutrophil chemokine pattern in tonsillitis.Clin Exp Immunol. 2004 Mar;135(3):511-8. doi: 10.1111/j.1365-2249.2003.02390.x.
|
|
|
|
|
|
|