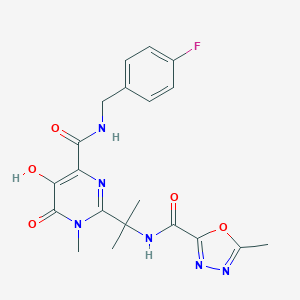
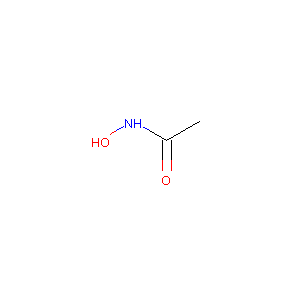
| 1 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
2007 FDA drug approvals: a year of flux. Nat Rev Drug Discov. 2008 Feb;7(2):107-9.
|
| 3 |
FDA Approved Drug Products from FDA Official Website. 2009. Application Number: (NDA) 018749.
|
| 4 |
Population pharmacokinetic analysis and pharmacogenetics of raltegravir in HIV-positive and healthy individuals. Antimicrob Agents Chemother. 2012 Jun;56(6):2959-66. doi: 10.1128/AAC.05424-11. Epub 2012 Feb 27.
|
| 5 |
Successful tacrolimus treatment following renal transplant in a HIV-infected patient with raltegravir previously treated with a protease inhibitor based regimen. Drug Metabol Drug Interact. 2011;26(3):139-41.
|
| 6 |
Exposure-related effects of atazanavir on the pharmacokinetics of raltegravir in HIV-1-infected patients. Ther Drug Monit. 2010 Dec;32(6):782-6.
|
| 7 |
Enzymatic, immunological and phylogenetic characterization of Brucella suis urease. BMC Microbiol. 2008 Jul 19;8:121.
|
| 8 |
New bispyridinium oximes: in vitro and in vivo evaluation of their biological efficiency in soman and tabun poisoning. Chem Biol Interact. 2008 Sep 25;175(1-3):413-6.
|
|
|
|
|
|
|