| 1 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
FDA Approved Drug Products from FDA Official Website. 2019. Application Number: (NDA) 019578
|
| 3 |
ClinicalTrials.gov (NCT04347031) An Open Randomized Study of the Effectiveness of Mefloquine for the Treatment of Patients With COVID19. U.S. National Institutes of Health.
|
| 4 |
Repurposing of clinically developed drugs for treatment of Middle East respiratory syndrome coronavirus infection. Antimicrob Agents Chemother. 2014 Aug;58(8):4885-93.
|
| 5 |
Mefloquine FDA Label
|
| 6 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7486).
|
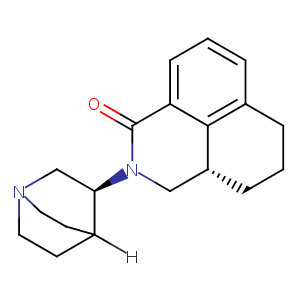
| 7 |
Palonosetron FDA Label
|
| 8 |
An ultrastructural study of the effects of mefloquine on malaria parasites. Am J Trop Med Hyg. 1987 Jan;36(1):9-14.
|
| 9 |
Polymorphisms in human MDR1 (P-glycoprotein): recent advances and clinical relevance. Clin Pharmacol Ther. 2004 Jan;75(1):13-33.
|
| 10 |
Effect of rifampin on plasma concentrations of mefloquine in healthy volunteers. J Pharm Pharmacol. 2000 Oct;52(10):1265-9.
|
| 11 |
Management of postoperative nausea and vomiting: focus on palonosetron. Ther Clin Risk Manag. 2009 Feb;5(1):21-34.
|
| 12 |
Pharmacokinetics, metabolism and excretion of intravenous [l4C]-palonosetron in healthy human volunteers. Biopharm Drug Dispos. 2004 Nov;25(8):329-37.
|
| 13 |
Cytochrome P450 2D6 metabolism and 5-hydroxytryptamine type 3 receptor antagonists for postoperative nausea and vomiting. Med Sci Monit. 2005 Oct;11(10):RA322-8.
|
| 14 |
Why are most phospholipidosis inducers also hERG blockers?. Arch Toxicol. 2017 Dec;91(12):3885-3895. doi: 10.1007/s00204-017-1995-9. Epub 2017 May 27.
|
|
|
|
|
|
|