Details of the Drug Combinations
General Information of This Drug (ID: DM1YBRM)
| Drug Name | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Ciclobenzaprina; Cyclobenz; Cyclobenzaprinum; Fexmid; Flexeril; Lisseril; Proeptatriene; Proheptatrien; Proheptatriene; Yurelax; Cyclobenzaprine [INN]; Flexeril hydrochloride; Proeptatriene [Italian]; MK 130; NCI78206; Amrix (TN); Ciclobenzaprina [INN-Spanish]; Cyclobenzaprine (INN); Cyclobenzaprinum [INN-Latin]; Fexmid(TN); Flexeril (TN); MK-130 HCl; Ro 4-1577; Apo-Cyclobenzaprine (TN); N,N-Dimethyl-3-(dibenzo(a,d)cycloheptene-4-ylidene)propylamine; N,N-Dimethyl-5H-dibenzo(a,d)cycloheptene-delta(sup 5),gamma-propylamine; N,N-dimethyl-5H-dibenzo(a,d)cycloheptene-Delta(5,gamma)-propylamine; 1-Propanamine, 3-(5H-dibenzo(a,d)cyclohepten-5-ylidene)-N,N-dimethyl-(9CI); 10,11-Dehydroamitriptyline; 3-(5H-Dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethyl-1-propanamine; 3-(5H-dibenzo[a,d][7]annulen-5-ylidene)-N,N-dimethylpropan-1-amine; 3-(5H-dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethylpropan-1-amine; 5-(3-Dimetilaminopropiliden)-5H-dibenzo-(a,d)-ciclopentene; 5-(3-Dimetilaminopropiliden)-5H-dibenzo-(a,d)-ciclopentene [Italian]; 9715 R.P; 9715 R.P.
|
||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||
| Therapeutic Class |
Antidepressants
|
||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||
| Structure |
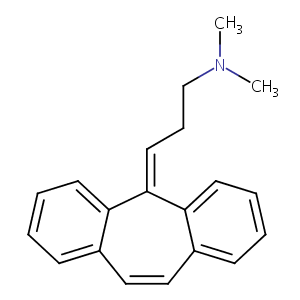 |
||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||
List of Combinatorial Drugs (CBD) Containing This Drug
|
2 Clinical Trial Drug Combination(s) Consisting of This drug
|
||||||||||||||||||||||||||||||
References
