Details of the Drug
General Information of Drug (ID: DM1YBRM)
| Drug Name |
Cyclobenzaprine
|
||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Ciclobenzaprina; Cyclobenz; Cyclobenzaprinum; Fexmid; Flexeril; Lisseril; Proeptatriene; Proheptatrien; Proheptatriene; Yurelax; Cyclobenzaprine [INN]; Flexeril hydrochloride; Proeptatriene [Italian]; MK 130; NCI78206; Amrix (TN); Ciclobenzaprina [INN-Spanish]; Cyclobenzaprine (INN); Cyclobenzaprinum [INN-Latin]; Fexmid(TN); Flexeril (TN); MK-130 HCl; Ro 4-1577; Apo-Cyclobenzaprine (TN); N,N-Dimethyl-3-(dibenzo(a,d)cycloheptene-4-ylidene)propylamine; N,N-Dimethyl-5H-dibenzo(a,d)cycloheptene-delta(sup 5),gamma-propylamine; N,N-dimethyl-5H-dibenzo(a,d)cycloheptene-Delta(5,gamma)-propylamine; 1-Propanamine, 3-(5H-dibenzo(a,d)cyclohepten-5-ylidene)-N,N-dimethyl-(9CI); 10,11-Dehydroamitriptyline; 3-(5H-Dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethyl-1-propanamine; 3-(5H-dibenzo[a,d][7]annulen-5-ylidene)-N,N-dimethylpropan-1-amine; 3-(5H-dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethylpropan-1-amine; 5-(3-Dimetilaminopropiliden)-5H-dibenzo-(a,d)-ciclopentene; 5-(3-Dimetilaminopropiliden)-5H-dibenzo-(a,d)-ciclopentene [Italian]; 9715 R.P; 9715 R.P.
|
||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||
| Therapeutic Class |
Antidepressants
|
||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||
| Structure |
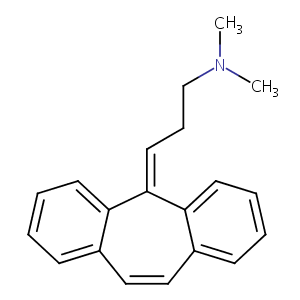 |
||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 275.4 | |||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 5.2 | ||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 3 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 0 | ||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 1 | ||||||||||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug-Metabolizing Enzyme (DME) |
|
||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||||||||||||
Drug-Drug Interaction (DDI) Information of This Drug
|
Coadministration of a Drug Treating the Same Disease as Cyclobenzaprine
Coadministration of a Drug Treating the Disease Different from Cyclobenzaprine (Comorbidity)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7152). | ||||
|---|---|---|---|---|---|
| 2 | Cyclobenzaprine FDA Label | ||||
| 3 | FDA Approved Drugs: Cyclobenzaprine | ||||
| 4 | BDDCS applied to over 900 drugs | ||||
| 5 | Cyclobenzaprine pharmacokinetics, including the effects of age, gender, and hepatic insufficiency. J Clin Pharmacol. 2002 Jan;42(1):61-9. doi: 10.1177/0091270002042001007. | ||||
| 6 | DPD Approved Drugs: Cyclobenzaprine | ||||
| 7 | Guerlavais V, Boeglin D, Mousseaux D, Oiry C, Heitz A, Deghenghi R, Locatelli V, Torsello A, Ghe C, Catapano F, Muccioli G, Galleyrand JC, Fehrentz JA, Martinez J: New active series of growth hormone secretagogues. J Med Chem. 2003 Mar 27;46(7):1191-203. | ||||
| 8 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 9 | Brioschi TM, Schramm SG, Kano EK, Koono EE, Ching TH, Serra CH, Porta V: Pharmacokinetics and bioequivalence evaluation of cyclobenzaprine tablets. Biomed Res Int. 2013;2013:281392. doi: 10.1155/2013/281392. Epub 2013 Sep 16. | ||||
| 10 | Torticollis under Cyclobenzaprine. Pharmacology. 2009 Jul 8;84(2):91-92. | ||||
| 11 | Identification of human liver cytochrome P450 isoforms involved in the in vitro metabolism of cyclobenzaprine. Drug Metab Dispos. 1996 Jul;24(7):786-91. | ||||
| 12 | Achamallah NS "Visual hallucinations after combining fluoxetine and dextromethorphan ." Am J Psychiatry 149 (1992): 1406. [PMID: 1530079] | ||||
| 13 | Boyer EW, Shannon M "The serotonin syndrome." N Engl J Med 352 (2005): 1112-20. [PMID: 15784664] | ||||
| 14 | Cohen MA, Alfonso CA, Mosquera M. Development of urinary retention during treatment with clozapine and meclizine [published correction appears in Am J Psychiatry 1994 Jun;151(6):952]. Am J Psychiatry. 1994;151(4):619-620. [PMID: 8147469] | ||||
| 15 | Ciraulo DA, Shader RI "Fluoxetine drug-drug interactions: I. Antidepressants and antipsychotics." J Clin Psychopharmacol 10 (1990): 48-50. [PMID: 1968472] | ||||
| 16 | Cerner Multum, Inc. "Australian Product Information.". | ||||
| 17 | US Food and Drug Administration "FDA warns about serious risks and death when combining opioid pain or cough medicines with benzodiazepines requires its strongest warning.". | ||||
| 18 | Kulik AV, Wilbur R "Delirium and stereotypy from anticholinergic antiparkinson drugs." Prog Neuropsychopharmacol Biol Psychiatry 6 (1982): 75-82. [PMID: 7202232] | ||||
| 19 | Beers MH, Ouslander JG, Rollingher I, Reuben DB, Brooks J, Beck JC "Explicit criteria for determining inappropriate medication use in nursing home residents." Arch Intern Med 151 (1991): 1825-32. [PMID: 1888249] | ||||
| 20 | Product Information. Ocaliva (obeticholic acid). Intercept Pharmaceuticals, Inc., New York, NY. | ||||
| 21 | Product Information. Tykerb (lapatinib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 22 | Multum Information Services, Inc. Expert Review Panel. | ||||
| 23 | Cole JM, Sheehan AH, Jordan JK "Concomitant use of ipratropium and tiotropium in chronic obstructive plmonary disease." Ann Pharmacother 46 (2012): 1717-21. [PMID: 23170031] | ||||
| 24 | US Food and Drug Administration "FDA warns about serious risks and death when combining opioid pain or cough medicines with benzodiazepines requires its strongest warning.". | ||||
| 25 | Product Information. Isturisa (osilodrostat). Recordati Rare Diseases Inc, Lebanon, NJ. | ||||
| 26 | Auclair B, Berning SE, Huitt GA, Peloquin CP "Potential interaction between itraconazole and clarithromycin." Pharmacotherapy 19 (1999): 1439-44. [PMID: 10600094] | ||||
| 27 | Product Information. Intelence (etravirine). Ortho Biotech Inc, Bridgewater, NJ. | ||||
| 28 | Product Information. Givlaari (givosiran). Alnylam Pharmaceuticals, Cambridge, MA. | ||||
| 29 | Product Information. Orladeyo (berotralstat). BioCryst Pharmaceuticals Inc, Durham, NC. | ||||
| 30 | Product Information. Suprep Bowel Prep Kit (magnesium/potassium/sodium sulfates). Braintree Laboratories, Braintree, MA. | ||||
| 31 | Cerner Multum, Inc. "UK Summary of Product Characteristics.". | ||||
| 32 | Product Information. Lorbrena (lorlatinib). Pfizer U.S. Pharmaceuticals Group, New York, NY. | ||||
| 33 | Product Information. Tagrisso (osimertinib). Astra-Zeneca Pharmaceuticals, Wilmington, DE. | ||||
| 34 | Product Information. Tabrecta (capmatinib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 35 | Product Information. Zelboraf (vemurafenib). Genentech, South San Francisco, CA. | ||||
| 36 | Product Information. Zulresso (brexanolone). Sage Therapeutics, Inc., Cambridge, MA. | ||||
| 37 | Product Information. Exjade (deferasirox). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 38 | Product Information. Addyi (flibanserin). Sprout Pharmaceuticals, Raleigh, NC. | ||||
| 39 | Product Information. Thalomid (thalidomide). Celgene Corporation, Warren, NJ. | ||||
| 40 | Alvine G, Black DW, Tsuang D "Case of delirium secondary to phenelzine/L-tryptophan combination." J Clin Psychiatry 51 (1990): 311. [PMID: 2365671] | ||||
| 41 | Product Information. Tasigna (nilotinib). Novartis Pharmaceuticals, East Hanover, NJ. | ||||
| 42 | Product Information. Wellbutrin XL (buPROPion). GlaxoSmithKline, Philadelphia, PA. | ||||
| 43 | EMA. European Medicines Agency. European Union "EMA - List of medicines under additional monitoring.". | ||||
| 44 | Product Information. Xeglyze (abametapir topical). Dr. Reddy's Laboratories Inc, Upper Saddle River, NJ. | ||||
| 45 | Product Information. Fycompa (perampanel). Eisai Inc, Teaneck, NJ. | ||||
| 46 | Product Information. Prograf (tacrolimus). Fujisawa, Deerfield, IL. | ||||
| 47 | Doherty MM, Charman WN "The mucosa of the small intestine: how clinically relevant as an organ of drug metabolism?" Clin Pharmacokinet 41 (2002): 235-53. [PMID: 11978143] | ||||
| 48 | Product Information. Tavalisse (fostamatinib). Rigel Pharmaceuticals, South San Francisco, CA. | ||||
