Details of the Drug Combinations
General Information of This Drug (ID: DMSQLZJ)
| Drug Name | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
NIFURTIMOX; Lampit; Bayer 2502; 23256-30-6; BAY 2502; Nifurtimox [INN:BAN]; Nifurtimoxum [INN-Latin]; CCRIS 2201; EINECS 245-531-0; CHEBI:7566; C10H13N3O5S; 4-Thiomorpholinamine, 3-methyl-N-((5-nitro-2-furanyl)methylene)-, 1,1-dioxide; 4-((5-Nitrofurfurylidene)amino)-3-methylthiomorpholine-1,1-dioxide; 3-Methyl-N-[(5-nitro-2-furanyl)methylene]-4-thiomorpholinamine 1,1-dioxide; 3-Methyl-4-(5'-nitrofurylidene-amino)-tetrahydro-4H-1,4-thiazine-1,1-dioxide
|
||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||
| Structure |
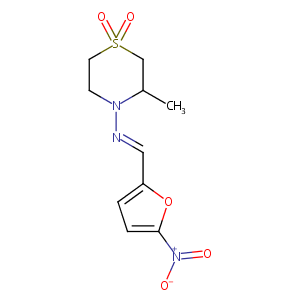 |
||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||
List of Combinatorial Drugs (CBD) Containing This Drug
|
1 Clinical Trial Drug Combination(s) Consisting of This drug
|
|||||||||||||||||||||||||
References
