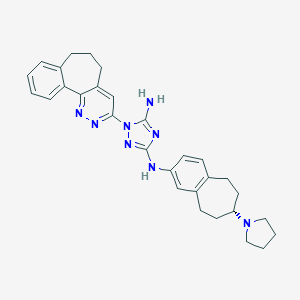
| Synonyms |
R428; 1037624-75-1; BGB324; R-428; BGB-324; UNII-0ICW2LX8AS; (S)-1-(6,7-dihydro-5H-benzo[6,7]cyclohepta[1,2-c]pyridazin-3-yl)-N3-(7-(pyrrolidin-1-yl)-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl)-1H-1,2,4-triazole-3,5-diamine; CHEMBL3809489; SYN1131; CS-1046; HY-15150; QC-11751; R 428; W-5845; Bemcentinib [USAN]; R428 (BGB324); Bemcentinib (USAN/INN); BemcentinibR428BGB324); SCHEMBL1639904; GTPL10478; BGB 324; DTXSID70673109; 1-(6,7-dihydro-5H-benzo[2,3]cyclohepta[2,4-d]pyridazin-3-yl)-3-N-[(7S)-7-pyrrolidin-1-yl-6,7,8,9-tetrahydro-5H-benzo[7]annulen-3-yl]-1,2,4-triazole-3,5-diamine; AMY16774; BCP21180; C30H34N8; EX-A1720; SYN-1131; BDBM50172079; NSC824183; s2841; WHO 10631; ZINC51951669; AKOS032947237; ACN-037541; DB12411; NSC-824183; SB16614; NCGC00386665-07; 1-(6,7-Dihydro-5H-benzo[6,7]cyclohepta[1,2-c]pyridazin-3-yl)-N~3~-[(7S)-7-(pyrrolidin-1-yl)-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl]-1H-1,2,4-triazole-3,5-diamine; 1H-1,2,4-Triazole-3,5-diamine, 1-(6,7-dihydro-5H-benzo(6,7)cyclohepta(1,2-C)pyridazin-3-yl)-N3-((7S)-6,7,8,9-tetrahydro-7-(1-pyrrolidinyl)-5H-benzocyclohepten-2-yl)-; AC-28444; AS-16270; KB-80319; D11438; Q27236818; BGB324; BGB-324; BGB 324; R 428; R-428; Bemcentinib; 1-(6,7-dihydro-5H-benzo[6,7]cyclohepta[1,2-c]pyridazin-3-yl)-N3-(7-(S)-(pyrrolidin-1-yl)-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-yl)-1H-1,2,4-triazole-3,5-diamine
|