Details of the Drug
General Information of Drug (ID: DM38CIV)
| Drug Name |
ELTANOLONE
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Pregnanolone; 3alpha-Hydroxy-5beta-pregnan-20-one; 128-20-1; Pregnanolone II; Pregnan-3alpha-ol-20-one; Eltanolona; Eltanolonum; 5beta-Pregnan-3alpha-ol-20-one; Eltanolone [INN]; SKF 6455; NSC 82867; UNII-BXO86P3XXW; Eltanolonum [INN-Latin]; Eltanolona [INN-Spanish]; Allopregnan-3 beta-ol-20-one; (3alpha,5beta)-3-hydroxypregnan-20-one; 3alpha-Hydroxy-5beta-tetrahydroprogesterone; 3alpha,5beta-Epimeric pregnanolone; Pregnanolone, (3alpha,5beta)-isomer; BXO86P3XXW
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
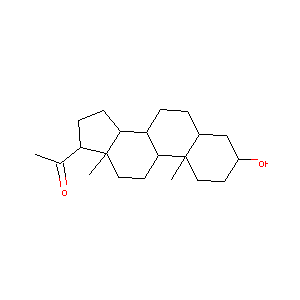 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 318.5 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 4.9 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 1 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 1 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 2 | ||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
References
