Details of the Drug
General Information of Drug (ID: DM3UQ9K)
| Drug Name |
Acolbifene
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Acolbifene; EM-652; 182167-02-8; UNII-815LJ9X0D1; 815LJ9X0D1; EM 652; Acolbifene [INN:BAN]; SCH 57068; AC1L4EAA; SCHEMBL406183; CHEMBL68055; CTK4F5320; SCH-57068; HY-16023A; ( )-(2S)-3-(4-Hydroxyphenyl)-4-methyl-2-(4-(2-(piperidin-1-yl)ethoxy)phenyl)-2H-1-benzopyran-7-ol; CS-0007143; (2S)-3-(4-hydroxyphenyl)-4-methyl-2-[4-(2-piperidin-1-ylethoxy)phenyl]-2H-chromen-7-ol; (S)-3-(4-hydroxyphenyl)-4-methyl-2-(4-(2-(piperidin-1-yl)ethoxy)phenyl)-2H-chromen-7-ol
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
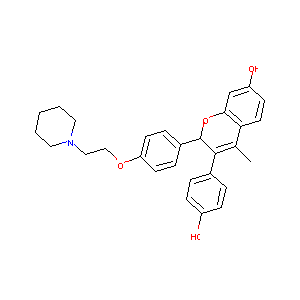 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 457.6 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 5.2 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 6 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
References
