Details of the Drug
General Information of Drug (ID: DME1AGO)
| Drug Name |
5-Fluoro-2'-Deoxyuridine-5'-Monophosphate
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
5-fluorodeoxyuridine monophosphate; FLUORODEOXYURIDYLATE; 5-fluoro-2'-deoxyuridine-5'-monophosphate; F-dUMP; 134-46-3; FdUMP; CHEMBL886; CHEBI:2129; UNII-7CJ707H131; 7CJ707H131; 88410-68-8; 5-FLUORO-2-DEOXYURIDINE MONOPHOSPHATE (FDUMP); [(2R,3S,5R)-5-(5-fluoro-2,4-dioxo-pyrimidin-1-yl)-3-hydroxy-tetrahydrofuran-2-yl]methyl dihydrogen phosphate; UFP; Poly(dF5U); Poly(5'-FU); Poly(5-fluoro-2'-deoxyuridylic acid); 5FdUMP; DB03761; 5'-Uridylic acid, 2'-deoxy-5-fluoro-; AC1L1RES; 5'-Uridylic acid,2'-deoxy-5-fluoro-, homopolymer
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
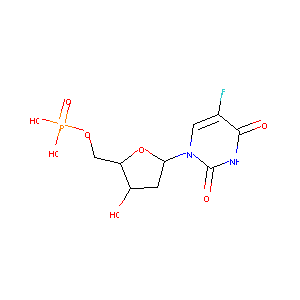 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 1 | Molecular Weight (mw) | 326.17 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | -2.8 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 4 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 4 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 9 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||||||||||||
References
