Details of the Drug
General Information of Drug (ID: DME9QGW)
| Drug Name |
Marizomib
|
||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
MARIZOMIB; salinosporamide A; 437742-34-2; (-)-Salinosporamide A; UNII-703P9YDP7F; NPI-0052; NPI 0052; ML 858; 703P9YDP7F; CHEBI:48045; (1R,4R,5S)-4-(2-chloroethyl)-1-[(S)-(1S)-cyclohex-2-en-1-yl(hydroxy)methyl]-5-methyl-6-oxa-2-azabicyclo[3.2.0]heptane-3,7-dione; Marizomib [USAN:INN]; marizomibum; Marizomib (USAN/INN); Salinosporamide A (NPI-0052, Marizomib); SCHEMBL151667; CHEMBL371405; NGWSFRIPKNWYAO-SHTIJGAHSA-N; ZINC3990364; BDBM50398608; 2531AH; AKOS027323566; DB11762; Z-3093; D09640; 855517-10-1
|
||||||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||||||
| Structure |
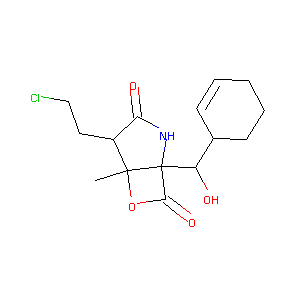 |
||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 313.77 | |||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 1.8 | ||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 4 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 4 | ||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
References
