Details of the Drug
General Information of Drug (ID: DMG2X3E)
| Drug Name |
Pyrantel
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
PYRANTEL; Pyrequan; 15686-83-6; Strongid; Pirantele [DCIT]; Pyrantelum; Pirantel; Pyrantel [INN:BAN]; Pyrantelum [INN-Latin]; Pirantel [INN-Spanish]; UNII-4QIH0N49E7; HSDB 3252; EINECS 239-774-1; CHEBI:8654; 4QIH0N49E7; (E)-1-Methyl-2-(2-(thiophen-2-yl)vinyl)-1,4,5,6-tetrahydropyrimidine; E-1,4,5,6-Tetrahydro-1-methyl-2-(2-(2-thienyl)vinyl)pyrimidine; Pirantele; 1-methyl-2-[(E)-2-(2-thienyl)vinyl]-5,6-dihydro-4H-pyrimidine; Pyrimidine, 1,4,5,6-tetrahydro-1-methyl-2-(2-(2-thienyl)vinyl)-, (E)-; Pin-X
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Affected Organisms |
Humans
|
||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
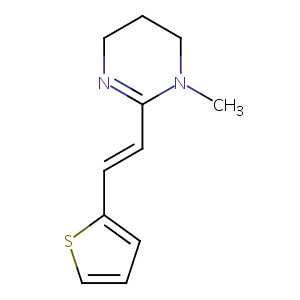 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 206.31 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 1.6 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 2 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 0 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 2 | ||||||||||||||||||||||
| ADMET Property |
|
||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
|---|---|---|---|---|---|
| 2 | BDDCS applied to over 900 drugs | ||||
| 3 | Pyrantel Pamoate Monograph | ||||
| 4 | The kinetic disposition of pyrantel citrate and pamoate and their efficacy against pyrantel-resistant Oesophagostomum dentatum in pigs. Int J Parasitol. 1996 Dec;26(12):1375-80. | ||||
| 5 | MSD vet manual | ||||
| 6 | Estimating the safe starting dose in phase I clinical trials and no observed effect level based on QSAR modeling of the human maximum recommended daily dose | ||||
| 7 | Identification of human cytochrome P(450)s that metabolise anti-parasitic drugs and predictions of in vivo drug hepatic clearance from in vitro data. Eur J Clin Pharmacol. 2003 Sep;59(5-6):429-42. | ||||
| 8 | Association of CYP1A1 and CYP1B1 inhibition in in vitro assays with drug-induced liver injury. J Toxicol Sci. 2021;46(4):167-176. doi: 10.2131/jts.46.167. | ||||
| 9 | Application of higher throughput screening (HTS) inhibition assays to evaluate the interaction of antiparasitic drugs with cytochrome P450s. Drug Metab Dispos. 2001 Jan;29(1):30-5. | ||||
