Details of the Drug
General Information of Drug (ID: DMJSKUI)
| Drug Name |
Norcisapride
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Norcisapride; 84946-16-7; CHEMBL1748; 4-Amino-5-chloro-2-methoxy-N-(3-methoxy-4-piperidyl)benzamide; 4-amino-5-chloro-2-methoxy-N-(3-methoxy-4-piperidinyl)benzamide; (4-Amino-5-chloro-2-methoxy)-N-[3-methoxy(4-piperidyl)]benzamide; 4-amino-5-chloro-2-methoxy-N-(3-methoxypiperidin-4-yl)benzamide; Benzamide,4-amino-5-chloro-2-methoxy-N-[(3S,4R)-3-methoxy-4-piperidinyl]-, hydrochloride(1:1); EINECS 284-619-3; AC1MI81F; SCHEMBL593405; CTK4E8653; OMLDMGPCWMBPAN-UHFFFAOYSA-N; BDBM50301927; AKOS030254741; API0006151; DB-076176
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
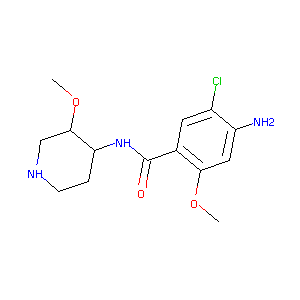 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 313.78 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 0.8 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 4 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 5 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
| Combinatorial Drugs (CBD) | Click to Jump to the Detailed CBD Information of This Drug | ||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
|||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug-Metabolizing Enzyme (DME) |
|
|||||||||||||||||||||||||||||||
 Drug Off-Target (DOT) |
|
|||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||||||||
Molecular Expression Atlas of This Drug
| The Studied Disease | Gastroesophageal reflux disease | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICD Disease Classification | DA22.Z | |||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Molecular Expression Atlas (MEA) | ||||||||||||||||||||||||||||||
References
