| 1 |
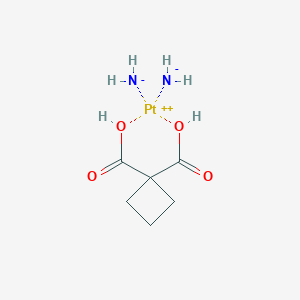
Carboplatin FDA Label
|
| 2 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7624).
|
| 3 |
Intravenous hydrophobic drug delivery: a porous particle formulation of paclitaxel (AI-850). Pharm Res. 2005 Mar;22(3):347-55.
|
| 4 |
ClinicalTrials.gov (NCT02944396) Study of Veliparib in Combination With Nivolumab and Platinum Doublet Chemotherapy in Participants With Metastatic or Advanced Non-Small Cell Lung Cancer (NSCLC)
|
| 5 |
ClinicalTrials.gov (NCT03400306) A Study Evaluating the Bioavailability and Food Effect of Veliparib Tablets Followed by an Extension in Subjects With Ovarian Cancer
|
| 6 |
ClinicalTrials.gov (NCT00993616) Belinostat and Carboplatin in Treating Patients With Recurrent or Persistent Ovarian Epithelial Cancer, Fallopian Tube Cancer, or Primary Peritoneal Cancer That Did Not Respond to Carboplatin or Cisplatin
|
| 7 |
ClinicalTrials.gov (NCT00077207) Carboplatin, Vincristine, and Temozolomide in Treating Children With Progressive and/or Symptomatic Low-Grade Glioma
|
| 8 |
ClinicalTrials.gov (NCT00002944) Combination Chemotherapy in Treating Children With Progressive Brain Tumors
|
| 9 |
ClinicalTrials.gov (NCT00362817) Carboplatin and Temozolomide (Temodar) for Recurrent and Symptomatic Residual Brain Metastases
|
| 10 |
ClinicalTrials.gov (NCT00230542) Carboplatin and Pemetrexed in Recurrent Platinum Sensitive Ovarian Cancer
|
| 11 |
ClinicalTrials.gov (NCT00101283) Pemetrexed Plus Gemcitabine or Carboplatin for Patients With Advanced Malignant Pleural Mesothelioma
|
| 12 |
ClinicalTrials.gov (NCT01478685) A Phase 1 Study of CC-486 as a Single Agent and in Combination With Carboplatin or ABI-007 in Subjects With Relapsed or Refractory Solid Tumors
|
| 13 |
ClinicalTrials.gov (NCT00387660) Irinotecan and Carboplatin in Treating Patients With Metastatic or Recurrent Small Cell Lung Cancer
|
| 14 |
ClinicalTrials.gov (NCT00002852) Surgery With or Without Chemotherapy in Treating Patients With Stage I Non-small Cell Lung Cancer
|
| 15 |
ClinicalTrials.gov (NCT00752011) Phase I Study of TAS-106 in Combo With Carboplatin
|
| 16 |
ClinicalTrials.gov (NCT00004859) Carboplatin, Paclitaxel, and Radiation Therapy With or Without Thalidomide in Patients With Stage III Non-small Cell Lung Cancer
|
| 17 |
ClinicalTrials.gov (NCT00036777) UCN-01 and Carboplatin in Treating Patients With Advanced Solid Tumors
|
| 18 |
ClinicalTrials.gov (NCT00005602) Radiation Therapy Plus Carboplatin and Lobradimil in Treating Children With Newly Diagnosed Brain Stem Gliomas
|
| 19 |
ClinicalTrials.gov (NCT00249964) Combination Paclitaxel, Carboplatin and Temozolomide
|
| 20 |
ClinicalTrials.gov (NCT01837862) A Phase I Study of Mebendazole for the Treatment of Pediatric Gliomas
|
| 21 |
ClinicalTrials.gov (NCT02312804) Ph Ib/BGJ398/Cervix and Other Solid Tumors
|
| 22 |
ClinicalTrials.gov (NCT00539331) Phase I/II Study of AZD2171 in Combination With Paclitaxel/Carboplatin in Japanese Non-Small Cell Lung Cancer Patients
|
| 23 |
ClinicalTrials.gov (NCT01000896) Study to Assess Safety and Tolerability of AZD0530 in Combination With Carboplatin and Paclitaxel
|
| 24 |
ClinicalTrials.gov (NCT01225302) A Study of Linifanib (ABT-869) in Combination With Carboplatin/Paclitaxel in Japanese Subjects With Non-Small Cell Lung Cancer (NSCLC)
|
| 25 |
ClinicalTrials.gov (NCT00408655) Temsirolimus, Carboplatin, and Paclitaxel in Treating Patients With Advanced Solid Tumors
|
| 26 |
ClinicalTrials.gov (NCT01249443) Paclitaxel and Carboplatin in Treating Patients With Metastatic or Recurrent Solid Tumors and HIV Infection
|
| 27 |
ClinicalTrials.gov (NCT00028587) PS-341 and Combination Chemotherapy in Treating Patients With Advanced Solid Tumors
|
| 28 |
ClinicalTrials.gov (NCT01256268) Carboplatin/Taxol/Ridaforolimus in Endometrial, Ovarian and Solids
|
| 29 |
ClinicalTrials.gov (NCT00529022) Azacitidine and Valproic Acid Plus Carboplatin in Patients With Ovarian Cancer
|
| 30 |
ClinicalTrials.gov (NCT00287937) Vorinostat, Paclitaxel, and Carboplatin in Treating Patients With Advanced or Refractory Solid Tumors
|
| 31 |
ClinicalTrials.gov (NCT00201734) Capecitabine, Carboplatin and Weekly Paclitaxel for Patients With Solid Tumors and Adenocarcinoma of Unknown Primary
|
| 32 |
ClinicalTrials.gov (NCT02039674) A Study of Pembrolizumab (MK-3475) in Combination With Chemotherapy or Immunotherapy in Participants With Non-small Cell Lung Cancer (MK-3475-021/KEYNOTE-021)
|
| 33 |
ClinicalTrials.gov (NCT02279992) Pilot Study of Vardenafil and Carboplatin in Patients With Gliomas and Brain Metastases
|
| 34 |
ClinicalTrials.gov (NCT03209401) Niraparib Plus Carboplatin in Patients With Homologous Recombination Deficient Advanced Solid Tumor Malignancies
|
| 35 |
ClinicalTrials.gov (NCT00581971) Radiosensitization With Celecoxib and Chemoradiation for Head and Neck Cancer
|
| 36 |
ClinicalTrials.gov (NCT00536601) High-Dose Chemotherapy With or Without Total-Body Irradiation Followed by Autologous Stem Cell Transplant in Treating Patients With Hematologic Cancer or Solid Tumors
|
| 37 |
ClinicalTrials.gov (NCT00090610) Second-Line Treatment for Patients With Platinum-Sensitive Ovarian Cancer
|
| 38 |
ClinicalTrials.gov (NCT00085358) Carboplatin and Paclitaxel With or Without Bevacizumab Compared to Docetaxel, Carboplatin, and Paclitaxel in Treating Patients With Stage II, Stage III, or Stage IV Ovarian Epithelial, Fallopian Tube, or Primary Peritoneal Cavity Carcinoma (Cancer)
|
| 39 |
ClinicalTrials.gov (NCT00714246) Bortezomib (PS-341) in Combination With Carboplatin and Docetaxel for Patients With Advanced Non-Small Cell Lung Cancer
|
| 40 |
ClinicalTrials.gov (NCT01372579) Carboplatin and Eribulin Mesylate in Triple Negative Breast Cancer Patients
|
| 41 |
ClinicalTrials.gov (NCT01795586) A Phase I Dose Escalation Study of Eribulin Plus Weekly Carboplatin for Metastatic Breast Patients
|
| 42 |
ClinicalTrials.gov (NCT00042835) Erlotinib and Radiation Therapy Plus Combination Chemotherapy in Treating Patients With Inoperable Stage III Non-Small Cell Lung Cancer
|
| 43 |
ClinicalTrials.gov (NCT01505569) Auto Transplant for High Risk or Relapsed Solid or CNS Tumors
|
| 44 |
ClinicalTrials.gov (NCT00017251) Combination Chemotherapy Plus Oblimersen in Treating Patients With Previously Untreated Extensive-Stage Small Cell Lung Cancer
|
| 45 |
ClinicalTrials.gov (NCT00027898) Bortezomib and Combination Chemotherapy in Treating Patients With Advanced Solid Tumors
|
| 46 |
ClinicalTrials.gov (NCT01127763) RAD001 Plus Carboplatin in Breast Cancer Patients
|
| 47 |
ClinicalTrials.gov (NCT01664754) Exemestane, Pemetrexed Disodium, and Carboplatin in Treating Post-Menopausal Women With Stage IV Non-Small Cell Lung Cancer
|
| 48 |
ClinicalTrials.gov (NCT00191451) A Study of Gemcitabine and Carboplatin (Plus Herceptin in Human Epidermal Growth Factor Receptor 2 Positive [HER2+] Patients) With Metastatic Breast Cancer
|
| 49 |
ClinicalTrials.gov (NCT02567799) BIO 300 Non-Small Cell Lung Cancer Study
|
| 50 |
ClinicalTrials.gov (NCT00502892) Topotecan, Ifosfamide and Carboplatin in Children and Young Adults With Solid Tumors
|
| 51 |
ClinicalTrials.gov (NCT00028561) BMS-247550 Plus Carboplatin in Treating Patients With Recurrent or Refractory Solid Tumors
|
| 52 |
ClinicalTrials.gov (NCT00077376) Trastuzumab, Ixabepilone, and Carboplatin in Treating Patients With HER2/Neu-Positive Metastatic Breast Cancer
|
| 53 |
ClinicalTrials.gov (NCT02050009) Metformin Hydrochloride, Carboplatin, and Paclitaxel in Treating Patients With Recurrent Ovarian, Fallopian Tube, or Primary Peritoneal Cancer
|
| 54 |
ClinicalTrials.gov (NCT00673179) Doxorubicin With Cisplatin, High-Dose Methotrexate, and Additional Risk-Adapted Outpatient Chemotherapy
|
| 55 |
ClinicalTrials.gov (NCT02813135) European Proof-of-Concept Therapeutic Stratification Trial of Molecular Anomalies in Relapsed or Refractory Tumors
|
| 56 |
ClinicalTrials.gov (NCT00005065) Chemotherapy, Radiation Therapy, and Surgery in Treating Patients With Stage IIIA Non-small Cell Lung Cancer
|
| 57 |
ClinicalTrials.gov (NCT00516724) Study to Assess the Safety and Tolerability of a PARP Inhibitor in Combination With Carboplatin and/or Paclitaxel
|
| 58 |
ClinicalTrials.gov (NCT02897375) Palbociclib With Cisplatin or Carboplatin in Advanced Solid Tumors
|
| 59 |
ClinicalTrials.gov (NCT01542047) A Combination of Pazopanib and Carboplatin in Advanced Solid Malignancies
|
| 60 |
ClinicalTrials.gov (NCT02941523) Safety, PK and Efficacy of NOX66 as a Monotherapy and Combined With Carboplatin in Refractory Solid Tumours
|
| 61 |
ClinicalTrials.gov (NCT03177291) Pirfenidone Combined With Standard First-Line Chemotherapy in Advanced-Stage Lung NSCLC
|
| 62 |
ClinicalTrials.gov (NCT00887575) Neoadjuvant Sunitinib With Paclitaxel/Carboplatin in Patients With Triple-Negative Breast Cancer
|
| 63 |
ClinicalTrials.gov (NCT01178112) Trientine and Carboplatin in Advanced Malignancies
|
| 64 |
ClinicalTrials.gov (NCT01636622) Study of Vemurafenib, Carboplatin, and Paclitaxel
|
| 65 |
ClinicalTrials.gov (NCT00095927) Randomized Amifostine For SCCHN
|
| 66 |
ClinicalTrials.gov (NCT00290537) Phase II Study of ZD6474 in Advanced NSCLC
|
| 67 |
ClinicalTrials.gov (NCT00288041) Bortezomib, Paclitaxel, and Carboplatin in Treating Patients With Metastatic Melanoma
|
| 68 |
ClinicalTrials.gov (NCT00473889) A Clinical Trial of Vorinostat (MK0683, SAHA) in Combination With FDA Approved Cancer Drugs in Patients With Advanced Non-Small Cell Lung Cancer (NSCLC)(0683-056)
|
| 69 |
ClinicalTrials.gov (NCT00006929) Suramin, Paclitaxel, and Carboplatin in Treating Patients With Stage IIIB or Stage IV Non-small Cell Lung Cancer
|
| 70 |
ClinicalTrials.gov (NCT01649947) Modulation of Autophagy in Patients With Advanced/Recurrent Non-small Cell Lung Cancer - Phase II
|
| 71 |
ClinicalTrials.gov (NCT00610714) AZD0530 Phase II Study in Patients With Advanced Ovarian Cancer
|
| 72 |
ClinicalTrials.gov (NCT02591615) Optimal Sequencing of Pembrolizumab (MK-3475) and Standard Platinum-based Chemotherapy in First-Line NSCLC
|
| 73 |
ClinicalTrials.gov (NCT02190695) Leukemia SPORE Phase II DAC Study for R/R and Elderly Acute AML and MDS
|
| 74 |
ClinicalTrials.gov (NCT01836679) Chidamide in Combination With Carboplatin and Paclitaxel in Advanced Non-small Cell Lung Cancer
|
| 75 |
ClinicalTrials.gov (NCT04771871) MicroRNA Profiles in Triple Negative Breast Cancer
|
| 76 |
ClinicalTrials.gov (NCT00003846) Radiation Therapy, Chemotherapy, and Peripheral Stem Cell Transplantation in Treating Patients With Primitive Neuroectodermal Tumors
|
| 77 |
ClinicalTrials.gov (NCT02670109) Autologous Hematopoietic Stem Cell Transplantation as Adjuvant Treatment for Triple Negative Breast Cancer Patients
|
| 78 |
ClinicalTrials.gov (NCT00828009) BLP25 Liposome Vaccine and Bevacizumab After Chemotherapy and Radiation Therapy in Treating Patients With Newly Diagnosed Stage IIIA or Stage IIIB Non-Small Cell LungCancer That Cannot Be Removed by Surgery. U.S. National Institutes of Health.
|
| 79 |
ClinicalTrials.gov (NCT00429182) Purged Circulating Tumor Cells (CTCs) From Metastatic Breast Cancer
|
| 80 |
ClinicalTrials.gov (NCT01344824) Carboplatin, Pemetrexed Disodium, and Bevacizumab for Patients With Stage III or IV Non-Small Cell Lung Cancer Who Are Light/Never Smokers
|
| 81 |
ClinicalTrials.gov (NCT00976677) Carboplatin, Paclitaxel, and Bevacizumab With or Without Erlotinib Hydrochloride in Treating Non-Smokers With Advanced Non-Small Cell Lung Cancer
|
| 82 |
ClinicalTrials.gov (NCT01014351) Study of Everolimus With Paclitaxel and Carboplatin in Patients With Metastatic Melanoma
|
| 83 |
ClinicalTrials.gov (NCT02531932) Comparison of Single-Agent Carboplatin vs the Combination of Carboplatin and Everolimus for the Treatment of Advanced Triple-Negative Breast Cancer
|
| 84 |
ClinicalTrials.gov (NCT00040794) Combination Chemotherapy, Radiation Therapy, and Gefitinib in Treating Patients With Stage III Non-Small Cell Lung Cancer
|
| 85 |
ClinicalTrials.gov (NCT00226590) Induction Gemcitabine & Carboplatin Followed by Paclitaxel & Carboplatin +XRT in NSCLC
|
| 86 |
ClinicalTrials.gov (NCT04464174) Ipatasertib Plus Non-Taxane Chemotherapy for Advanced or Metastatic Triple-Negative Breast Cancer
|
| 87 |
ClinicalTrials.gov (NCT02272790) Adavosertib Plus Chemotherapy in Platinum-Resistant Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer
|
| 88 |
ClinicalTrials.gov (NCT04038502) Carboplatin or Olaparib for BRcA Deficient Prostate Cancer
|
| 89 |
ClinicalTrials.gov (NCT00695292) Irinotecan, Carboplatin, and Sunitinib in First Line Extensive-Stage Small Cell Lung Cancer
|
| 90 |
ClinicalTrials.gov (NCT03656393) To Evaluate the Efficacy and Safety of Gefitinib in Adjuvant Chemotherapy for Lung Adenocarcinoma
|
| 91 |
ClinicalTrials.gov (NCT03850444) Study of Pembrolizumab (MK-3475) Versus Platinum-Based Chemotherapy for Participants With Programmed Cell Death-Ligand 1 (PD-L1)-Positive Advanced or Metastatic Non-Small Cell Lung Cancer (MK-3475-042/KEYNOTE-042)-China Extension Study
|
|
|
|
|
|
|