Details of the Drug
General Information of Drug (ID: DMWIO46)
| Drug Name |
Camphor
|
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
camphor; 76-22-2; 2-Camphanone; 2-Bornanone; DL-Camphor; (+)-Camphor; l-(-)-Camphor; (+/-)-Camphor; Root bark oil; 1,7,7-Trimethylbicyclo[2.2.1]heptan-2-one; Bornan-2-one; Alphanon; Kampfer; Gum camphor; Spirit of camphor; Formosa camphor; Laurel camphor; 2-Camphonone; Matricaria camphor; Camphor, synthetic; Bornane, 2-oxo-; 1,7,7-Trimethylnorcamphor; Root bark spirit; 2-Keto-1,7,7-trimethylnorcamphane; DL-Bornan-2-one; D-(+)-Camphor; Norcamphor, 1,7,7-trimethyl-; 21368-68-3; Camphor, (1R,4R)-(+)-; Bicyclo[2.2.1]heptan-2-one, 1,7,7-tri
|
||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||
| Structure |
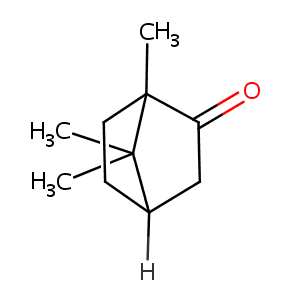 |
||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 152.23 | |||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 2.2 | ||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 0 | ||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 0 | ||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 1 | ||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug-Metabolizing Enzyme (DME) |
|
||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug Off-Target (DOT) |
|
||||||||||||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||||||||||||
References
