| 1 |
ClinicalTrials.gov (NCT01088048) Study to Investigate Idelalisib in Combination With Chemotherapeutic Agents, Immunomodulatory Agents and Anti-CD20 Monoclonal Antibody (mAb) in Participants With Relapsed or Refractory Indolent B-cell Non-Hodgkin's Lymphoma, Mantle Cell Lymphoma or Chronic Lymphocytic Leukemia
|
| 2 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6741).
|
| 3 |
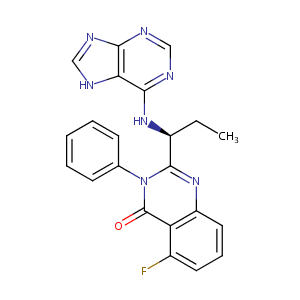
Idelalisib FDA Label
|
| 4 |
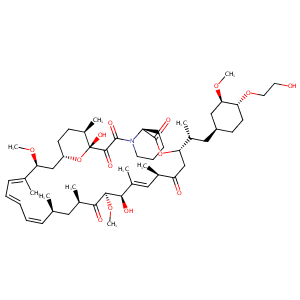
Everolimus FDA Label
|
| 5 |
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015
|
| 6 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 5889).
|
| 7 |
Coronaviruses - drug discovery and therapeutic options. Nat Rev Drug Discov. 2016 May;15(5):327-47.
|
| 8 |
2014 FDA drug approvals. Nat Rev Drug Discov. 2015 Feb;14(2):77-81.
|
| 9 |
DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res. 2018 Jan 4;46(D1):D1074-D1082. (ID: DB09054)
|
| 10 |
FDA label of Idelalisib. The 2020 official website of the U.S. Food and Drug Administration.
|
| 11 |
PI3K/AKT inhibitors aggravate death receptor-mediated hepatocyte apoptosis and liver injury. Toxicol Appl Pharmacol. 2019 Oct 15;381:114729. doi: 10.1016/j.taap.2019.114729. Epub 2019 Aug 22.
|
| 12 |
Roles of pulmonary telocytes in airway epithelia to benefit experimental acute lung injury through production of telocyte-driven mediators and exosomes. Cell Biol Toxicol. 2023 Apr;39(2):451-465. doi: 10.1007/s10565-021-09670-5. Epub 2022 Jan 3.
|
| 13 |
Induction of prolonged early G1 arrest by CDK4/CDK6 inhibition reprograms lymphoma cells for durable PI3K inhibition through PIK3IP1. Cell Cycle. 2013 Jun 15;12(12):1892-900. doi: 10.4161/cc.24928. Epub 2013 May 15.
|
| 14 |
Regulatory roles of NAT10 in airway epithelial cell function and metabolism in pathological conditions. Cell Biol Toxicol. 2023 Aug;39(4):1237-1256. doi: 10.1007/s10565-022-09743-z. Epub 2022 Jul 25.
|
| 15 |
Mammalian target of rapamycin, its mode of action and clinical response in metastatic clear cell carcinoma. Gan To Kagaku Ryoho. 2009 Jul;36(7):1076-9.
|
| 16 |
Closer to the Site of Action: Everolimus Concentrations in Peripheral Blood Mononuclear Cells Correlate Well With Whole Blood Concentrations. Ther Drug Monit. 2015 Oct;37(5):675-80.
|
| 17 |
The evolving experience using everolimus in clinical transplantation. Transplant Proc. 2004 Mar;36(2 Suppl):495S-499S.
|
|
|
|
|
|
|