| 1 |
Loss of function mutations in VARS encoding cytoplasmic valyl-tRNA synthetase cause microcephaly, seizures, and progressive cerebral atrophy.Hum Genet. 2018 Apr;137(4):293-303. doi: 10.1007/s00439-018-1882-3. Epub 2018 Apr 24.
|
| 2 |
Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015
|
| 3 |
FDA Approved Drug Products from FDA Official Website. 2019. Application Number: (NDA) 019578
|
| 4 |
ClinicalTrials.gov (NCT04347031) An Open Randomized Study of the Effectiveness of Mefloquine for the Treatment of Patients With COVID19. U.S. National Institutes of Health.
|
| 5 |
Repurposing of clinically developed drugs for treatment of Middle East respiratory syndrome coronavirus infection. Antimicrob Agents Chemother. 2014 Aug;58(8):4885-93.
|
| 6 |
Mefloquine FDA Label
|
| 7 |
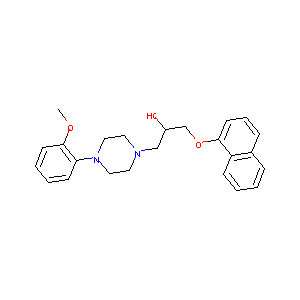
Pharmacophore identification of alpha(1A)-adrenoceptor antagonists. Bioorg Med Chem Lett. 2005 Feb 1;15(3):657-64.
|
| 8 |
Identification of human cytochrome P450 isozymes involved in the metabolism of naftopidil enantiomers in vitro. J Pharm Pharmacol. 2014 Nov;66(11):1534-51.
|
| 9 |
Autophagy Induced by Naftopidil Inhibits Apoptosis of Human Gastric Cancer Cells. Anticancer Res. 2018 Feb;38(2):803-809. doi: 10.21873/anticanres.12287.
|
| 10 |
An ultrastructural study of the effects of mefloquine on malaria parasites. Am J Trop Med Hyg. 1987 Jan;36(1):9-14.
|
| 11 |
Polymorphisms in human MDR1 (P-glycoprotein): recent advances and clinical relevance. Clin Pharmacol Ther. 2004 Jan;75(1):13-33.
|
| 12 |
Effect of rifampin on plasma concentrations of mefloquine in healthy volunteers. J Pharm Pharmacol. 2000 Oct;52(10):1265-9.
|
|
|
|
|
|
|