Details of the Drug Combination
General Information of Drug Combination (ID: DCTYYX4)
| Drug Combination Name |
Tenofovir disoproxil GS-9620
|
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Indication |
|
|||||||||||||||||
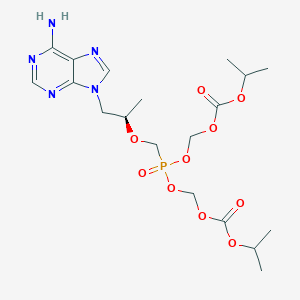
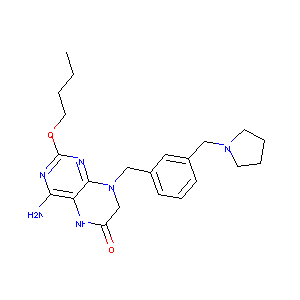
| Component Drugs | Tenofovir disoproxil | GS-9620 | ||||||||||||||||
| N.A. | Small molecular drug | |||||||||||||||||
|
|
|||||||||||||||||
| 2D MOL | 2D MOL | |||||||||||||||||
| 3D MOL is unavailable | 3D MOL | |||||||||||||||||
Molecular Interaction Atlas of This Drug Combination
| Molecular Interaction Atlas (MIA) | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Tenofovir disoproxil Interacts with 1 DTP Molecule(s)
|
||||||||||||||||||||||||||
|
Tenofovir disoproxil Interacts with 1 DME Molecule(s)
|
||||||||||||||||||||||||||
| Indication(s) of GS-9620 |
|
|||||||||||||||||||||||||
|
GS-9620 Interacts with 1 DTT Molecule(s)
|
||||||||||||||||||||||||||
|
GS-9620 Interacts with 1 DME Molecule(s)
|
||||||||||||||||||||||||||
References
