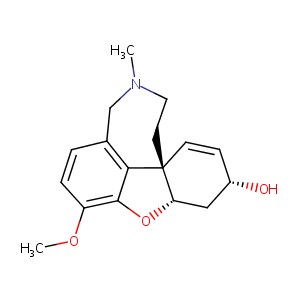
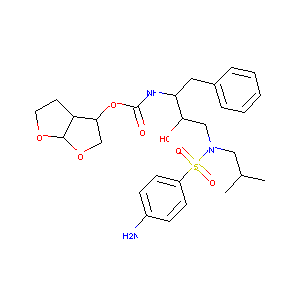
| 1 |
Recurrent recessive mutation in deoxyguanosine kinase causes idiopathic noncirrhotic portal hypertension.Hepatology. 2016 Jun;63(6):1977-86. doi: 10.1002/hep.28499. Epub 2016 Mar 31.
|
| 2 |
URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6693).
|
| 3 |
Galantamine FDA Label
|
| 4 |
Natural products as sources of new drugs over the last 25 years. J Nat Prod. 2007 Mar;70(3):461-77.
|
| 5 |
Clinical pipeline report, company report or official report of the Pharmaceutical Research and Manufacturers of America (PhRMA)
|
| 6 |
[From symptomatic to disease modifying therapy Recent developments in the pharmacotherapy of Alzheimer's disease]. Fortschr Neurol Psychiatr. 2009 Jun;77(6):326-33.
|
| 7 |
Clinical pharmacokinetics of galantamine. Clin Pharmacokinet. 2003;42(15):1383-92.
|
| 8 |
Lichens of parmelioid clade as promising multitarget neuroprotective agents. Chem Res Toxicol. 2019 Jun 17;32(6):1165-1177.
|
| 9 |
Inhibition of human carboxylesterases hCE1 and hiCE by cholinesterase inhibitors. Chem Biol Interact. 2013 Mar 25;203(1):226-30.
|
| 10 |
Cholinergic drugs potentiate human nicotinic alpha4beta2 acetylcholine receptors by a competitive mechanism. Eur J Pharmacol. 2005 Feb 21;509(2-3):97-108. doi: 10.1016/j.ejphar.2004.12.037.
|
| 11 |
Potencies and selectivities of inhibitors of acetylcholinesterase and its molecular forms in normal and Alzheimer's disease brain. Acta Biol Hung. 2003;54(2):183-9. doi: 10.1556/ABiol.54.2003.2.7.
|
| 12 |
2006 drug approvals: finding the niche. Nat Rev Drug Discov. 2007 Feb;6(2):99-101.
|
| 13 |
Impact of drug transporters on cellular resistance towards saquinavir and darunavir. J Antimicrob Chemother. 2010 Nov;65(11):2319-28.
|
| 14 |
Organic anion transporting polypeptide 1B1: a genetically polymorphic transporter of major importance for hepatic drug uptake. Pharmacol Rev. 2011 Mar;63(1):157-81.
|
| 15 |
P-glycoprotein mediates efflux transport of darunavir in human intestinal Caco-2 and ABCB1 gene-transfected renal LLC-PK1 cell lines. Biol Pharm Bull. 2009 Sep;32(9):1588-93.
|
| 16 |
Drug interactions with new and investigational antiretrovirals. Clin Pharmacokinet. 2009;48(4):211-41.
|
|
|
|
|
|
|