Details of the Drug
General Information of Drug (ID: DMC5TV6)
| Drug Name |
TPI-287
|
||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
849213-15-6; (1S,2S,4S,7S,7aR,7a1S,10aS,11aR,13aS,13bR)-1-(benzoyloxy)-4-(((2R,3S)-3-((tert-butoxycarbonyl)amino)-2-hydroxy-5-methylhexanoyl)oxy)-2-hydroxy-5,7a1,14,14-tetramethyl-9-vinyl-2,3,4,7,7a,7a1,10a,11,11a,13,13a,13b-dodecahydro-1H-8,10,12-trioxa-2,6-methanocyclobuta[b]cyclodeca[de]naphthalene-7,13a-diyl diacetate; TPI 287; SCHEMBL10000720; DTXSID50233967; Q27273546
|
||||||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||||||
| Structure |
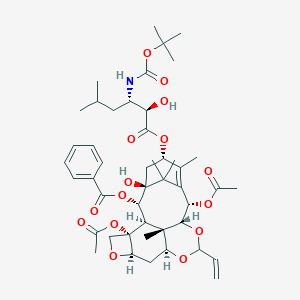 |
||||||||||||||||||||||||||||||||||
| 3D MOL is unavailable | 2D MOL | ||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 3 | Molecular Weight (mw) | 870 | |||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 4.6 | ||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 17 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 3 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 15 | ||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
References
