Details of the Drug
General Information of Drug (ID: DMHCI9S)
| Drug Name |
Flurandrenolide
|
||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms |
Cordran; Drenison; Drocort; Floudroxycortide; Fludrossicortide; Fludroxicortida; Fludroxicortidum; Fludroxycortide; Fludroxycortidum; Fluorandrenolone; Fluradrenolide; Flurandrenolone; Haelan; Sermaka; CORDRAN SP; Fludrossicortide [DCIT]; Fluorandrenolone acetonide; Flurandrenolide [USAN]; Flurandrenolone acetonide; L 33379; Alondra-F; CORDRAN (TN); Cordran tape (TN); Fludroxicortida [INN-Spanish]; Fludroxycortidum [INN-Latin]; Flurandrenolide (USP); Haldrone-F; Fludroxycortide (JAN/INN); Acetonide of 6alpha-fluoro-16alpha-hydroxyhydrocortisone; Pregn-4-ene-3,20-dione, 6alpha-fluoro-11beta,16alpha,17,21-tetrahydroxy-, cyclic 16,17-acetal with acetone; Pregn-4-ene-3,20-dione, 6-alpha-fluoro-11-beta,16-alpha,17,21-tetrahydroxy-, cyclic 16,17-acetal with acetone; 6alpha-Fluoro-11beta,16alpha,17,21-tetrahydroxypregn-4-ene-3,20-dione, cyclic 16,17-acetal with acetone; 6alpha-Fluoro-11beta,16alpha,17,21-tetrahydroxyprogesterone cyclic 16,17-acetal with acetone; 6alpha-Fluoro-16alpha-hydroxyhydrocortisone 16,17-acetonide
|
||||||||||||||||||||||||||||||||||
| Indication |
|
||||||||||||||||||||||||||||||||||
| Therapeutic Class |
Antiinflammatory Agents
|
||||||||||||||||||||||||||||||||||
| Affected Organisms |
Humans and other mammals
|
||||||||||||||||||||||||||||||||||
| ATC Code | |||||||||||||||||||||||||||||||||||
| Drug Type |
Small molecular drug
|
||||||||||||||||||||||||||||||||||
| Structure |
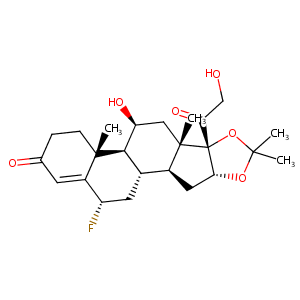 |
||||||||||||||||||||||||||||||||||
| 3D MOL | 2D MOL | ||||||||||||||||||||||||||||||||||
| #Ro5 Violations (Lipinski): 0 | Molecular Weight (mw) | 436.5 | |||||||||||||||||||||||||||||||||
| Logarithm of the Partition Coefficient (xlogp) | 1.4 | ||||||||||||||||||||||||||||||||||
| Rotatable Bond Count (rotbonds) | 2 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Donor Count (hbonddonor) | 2 | ||||||||||||||||||||||||||||||||||
| Hydrogen Bond Acceptor Count (hbondacc) | 7 | ||||||||||||||||||||||||||||||||||
| ADMET Property | |||||||||||||||||||||||||||||||||||
| Chemical Identifiers |
|
||||||||||||||||||||||||||||||||||
| Cross-matching ID | |||||||||||||||||||||||||||||||||||
| Repurposed Drugs (RPD) | Click to Jump to the Detailed RPD Information of This Drug | ||||||||||||||||||||||||||||||||||
Molecular Interaction Atlas of This Drug
 Drug Therapeutic Target (DTT) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Drug-Metabolizing Enzyme (DME) |
|
||||||||||||||||||||||||||
| Molecular Interaction Atlas (MIA) | |||||||||||||||||||||||||||
Drug Inactive Ingredient(s) (DIG) and Formulation(s) of This Drug
References
| 1 | Flurandrenolide FDA Label | ||||
|---|---|---|---|---|---|
| 2 | URL: http://www.guidetopharmacology.org Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7606). | ||||
| 3 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. | ||||
| 4 | Prediction of cytochrome P450 isoform responsible for metabolizing a drug molecule. BMC Pharmacol. 2010 Jul 16;10:8. | ||||
